 |
 |
| Plant Pathol J > Volume 38(5); 2022 > Article |
|
Abstract
Erwinia amylovora is a causative pathogen of fire blight disease, affecting apple, pear, and other rosaceous plants. Currently, management of fire blight relies on cultural and chemical practices, whereas it has been known that few biological resources exhibit disease control efficacy against the fire blight. In the current study, we found that an SFC20201208-M01 fungal isolate exhibits antibacterial activity against E. amylovora TS3128, and the isolate was identified as a Penicillium brasilianum based on the β-tubulin (BenA) gene sequence. To identify active compounds from the P. brasilianum culture, the culture filtrate was partitioned with ethyl acetate and n-butanol sequentially. From the ethyl acetate layer, we identified two new compounds (compounds 3-4) and two known compounds (compounds 1-2) based on spectroscopic analyses and comparison with literature data. Of these active compounds, penicillic acid (1) exhibited promising antibacterial activity against E. amylovora TS3128 with a minimal inhibitory concentration value of 25 μg/ml. When culture filtrate and penicillic acid (125 μg/ml) were applied onto Chinese pearleaf crab apple seedlings prior to inoculation of E. amylovora TS3128, the development of fire blight disease was effectively suppressed in the treated plants. Our results provide new insight into the biocontrol potential of P. brasilianum SFC20201208-M01 with an active ingredient to control fire blight.
Erwinia amylovora is a Gram-negative bacterial pathogen and belongs to the Enterobacterales (Hauben and Swings, 2005). This pathogen causes a devastating disease, fire blight on plants belonging to the family Rosaceae, such as apples and pears, which results in severe damage to crop quality and quantity of fruit yield (Malnoy et al., 2012; Norelli et al., 2003). E. amylovora can infect the tree trunk, shoots, flowers, and natural wounds through abiotic and biotic vectors, and the disease symptoms such as necrotic and blackened lesions, bacterial ooze, and tree canker can be found in various parts of infected trees, including blossoms, fruits, and branches (Kong et al., 2021; Vanneste, 2000; Vrancken et al., 2013). Fire blight disease was firstly found in New York in the late 1700s on Rosaceae plants and had been broken out in North America, European, and Asia (Erfani et al., 2013; Schroth et al., 1974; Sobiczewski et al., 2015). In Korea, the fire blight disease was firstly confirmed in 2015, and then this disease has continued to spread out to new regions (Park et al., 2016).
Considering that E. amylovora is highly infectious and quickly disseminated by various environmental factors such as wind, rain, pollen, or insect vectors, it is challenging to control E. amylovora (Aćimović et al., 2015; Vanneste, 2000). Management of fire blight has typically depended on cultural and chemical controls (Norelli et al., 2003). However, the use of the chemicals such as copper-based bactericides and antibiotics has caused a high risk of phytotoxicity or the emergence of resistant pathogens (Aćimović et al., 2015; McManus et al., 2002; Sholberg et al., 2001). Given that resistant strains against antibiotics have been reported in the United States, Canada, New Zealand, Israel, and Mexico, the use of antibiotics has been limited in the field (Coyier and Covey, 1975; De León Door et al., 2013; Russo et al., 2008; Thomson et al., 1993). To compensate for the shortcomings of chemicals including antibiotics, biological controls using beneficial microbes have been considered as the most eco-friendly and sustainable control method. Although Bacillus subtilis, Lactobacillus plantarum, Pseudomonas fluorescens, and Pantoea agglomerans have been reported as biological control agents to manage fire blight disease, there is still a need for the discovery of valuable microbes with a promising disease control efficacy for the fire blight (Bonaterra et al., 2007; Broggini et al., 2005; Dagher et al., 2020; Pusey et al., 2011; Roselló et al., 2013).
The marine environment has been investigated for new natural resources containing bioactive compounds with benefits for humans, animals, and plants (Hamed et al., 2015). In particular, marine-derived fungi have been considered as a rich source of secondary metabolites with promising antimicrobial effects (Naglot et al., 2015). During the screening procedure with the culture filtrates of marine-derived fungi showing antibacterial activity, we found that a strain SFC20201208-M01 isolated from the foreshore soil has promising antibacterial activity against E. amylovora. Therefore, this study aimed to isolate and identify active metabolites and investigate in vitro and in vivo antibacterial activities of SFC20201208-M01 and its metabolites against E. amylovora.
A bacterial pathogen E. amylovora TS3128 provided by Rural Development Administration (Wanju, Korea) was maintained on mannitol glutamate yeast extract medium (MGY; d-mannitol 10 g, L-glutamic acid 2 g, KH2PO4 0.5 g, NaCl 0.2 g, MgSO4·7H2O 0.2 g, yeast extract 1 g per liter) at 28°C. A fungus SFC20201208-M01 was kindly provided by the Marine Fungal Resource Bank of Seoul National University, which was isolated from the foreshore soil of Jangheungri, Incheon, Korea. The fungus was maintained on potato dextrose agar (BD Difco, Sparks, MD, USA) medium at 25°C. For phenotypic observation and antibacterial activity assay, the fungus SFC20201208-M01 was grown on malt extract broth medium (MEB; malt extract 6 g, maltose 1.8 g, dextrose 6 g, yeast extract 1.2 g per liter), Sabouraud’s dextrose broth medium (SDB; dextrose 20 g, mycological peptone 10 g per liter), and Czapek-Dox broth medium (CDB; sucrose 30 g, NaNO3 3 g, K2HPO4 1 g, MgSO4 0.5 g, KCl 0.5 g, FeSO4 0.01 g per liter) supplemented with 1.5% of agar as needed. To isolate active compounds, SFC20201208-M01 was grown in potato dextrose broth (PDB; BD Difco) medium at 25°C for 14 days.
To isolate the genomic DNA (gDNA) of SFC20201208-M01, the fungus was grown in 50 ml of PDB medium at 25°C for 4 days on a rotary shaker. The gDNA was extracted using the cetyltrimethylammonium bromide procedure as previously described (Oh et al., 2016). For phylogenetic analysis, the β-tubulin (BenA) gene was amplified by primer set bt2a (5′-GGTAACCAAATCGGTGCTGCTTTC-3′) and bt2b (5′-ACCCTCAGTGTAGTGACCCTTGGC-3′) (Glass and Donaldson, 1995). The resulting amplicon was purified using the GeneAll ExpinTM PCR purification kit (GeneAll, Seoul, Korea) and then analyzed using the corresponding PCR primers by Macrogen (Seoul, Korea). The resulting sequence was analyzed with the BLASTn program of the NCBI (http://www.ncbi.nlm.nih.gov). The sequences were aligned using ClustalW implemented in MEGA version 7, and distances were estimated based on the model. A phylogenetic tree was generated using the neighbor-joining method with 1,000 bootstrap analyses (Saitou and Nei, 1987).
To isolate the active compounds from the SFC20201208-M01 culture filtrate, 20 agar plugs punched by a 6-mm-diameter cork-borer from SFC20201208-M01 culture plates were inoculated into 400 ml of PDB. The 14-day-old culture filtrate (4.6 l) of SFC20201208-M01 was partitioned with organic solvents ethyl acetate (EtOAc) and n-butanol (n-BuOH) sequentially. Based on the result of in vitro antibacterial activity, the active EtOAc extract (4.5 g) was applied to a SNAP KP-Sil 50 g cartridge in a medium pressure liquid chromatography system (Isolera One, Biotage, Uppsala, Sweden) and eluted with a gradient mixture of methylene chloride/EtOAc (90:10 to 70:30, v/v) to give a main compound 1 (769.9 mg) and other fractions E5 and E6 (75.2 mg and 101.9 mg) containing minor compounds. These fractions were pooled and separated by a preparative high-performance liquid chromatography (HPLC) using an LC-20AR system (Shimadzu, Kyoto, Japan) equipped with a Capcell Pak C18 column (20 × 250 mm i.d., 5 μm; Shiseido Co., Ltd., Tokyo, Japan) eluting with a gradient mixture of aqueous methanol (10-20%, v/v, at a flow rate of 6 ml/min in 60 min) to yield compounds 2 (52.7 mg, tR = 28.3 min), 3 (3.4 mg, tR = 28.9 min), and 4 (3.5 mg, tR = 35.3 min).
The chemical structures of the purified compounds were determined by spectroscopic analyses and comparisons with values in previous literature. High-resolution electrospray ionization mass spectrometry (HRESIMS) data were determined by a Synapt G2 system (Waters Co., Milford, MA, USA). The 1D and 2D nuclear magnetic resonance (NMR) spectra were recorded by a Bruker Advance 400 MHz spectrometer (Burker BioSpin, Rheinstetten, Germany) at 400 MHz for 1H and 100 MHz for 13C in methanol-d4 (Cambridge Isotope Laboratories, Tewksbury, MA, USA). Chemical shifts were referenced to the solvent peaks (δH 4.87 and δC 49.0 for methanol-d4).
The minimal inhibitory concentration (MIC) and half-maximal inhibitory concentration (IC50) values for E. amylovora TS3128 were determined by the two-fold serial dilution assay as described previously (Espinel-Ingroff et al., 2005). Briefly, a bacterial suspension (2 × 105 cfu/ml of MGY) of E. amylovora TS3128 was added to the wells of a 96-well microtiter plate. The culture filtrates were added to the well at an initial concentration of 20% and then serially two-fold diluted. The purified compounds dissolved in dimethyl sulfoxide (DMSO) were added and then serially two-fold diluted to reach the final concentrations ranging from 0.1 to 200 μg/ml; the final concentration of DMSO did not exceed 1%. The antibiotic streptomycin sulfate (Sigma-Aldrich, St. Louis, MO, USA) and an MGY medium containing 1% DMSO were used as positive and negative controls, respectively. The microtiter plates were incubated for 24 h at 28°C, and the MIC values were determined by visual inspection of complete growth inhibition. IC50 values was calculated as follows: [1 - (OD600 of treatment/OD600 of control) × 100]. The inhibitory effects on the bacterial growth were also visualized by Prestoblue solution (Thermo Fisher Scientific, Waltham, MA, USA) according to the manufacturer’s instructions. The assay was performed two times with three replicates at all concentrations investigated.
To evaluate the disease control efficacy of the SFC20201208-M01 culture filtrate and its metabolites against fire blight disease, Chinese pearleaf crab apple seedlings (Malus asiatica Nakai) were used as a host plant. Briefly, the Chinese pearleaf crab apple seeds were sown and grown in a greenhouse at 25 ± 5°C for 4-5 weeks. One day before inoculation of the pathogen, the culture filtrate was directly applied onto the plant by spraying. In the case of an active compound, the plants were also treated with the pure compound (125, 250, and 500 μg/ml) dissolved in 1% DMSO (v/v), using the same method for the culture filtrates. When the culture filtrates and pure compounds were applied to the plants, the samples contained 0.025% Tween 20 (w/v) as a wetting agent. Streptomycin (100 μg/ml) and 1% DMSO were used as positive and negative controls, respectively. The treated plants were inoculated with a bacterial suspension (4 × 108 cfu/ml) of E. amylovora TS3128 grown MGY broth. The inoculated plants were incubated for 2 days in a humidified chamber (25°C) and then moved to a growth chamber (25°C, 80% relative humidity, and 12-h photoperiod). After 7 days of additional incubation, we investigated the disease severity determined by the modified Wang methods (Wang et al., 2010). The disease severity index (DSI) ranged from 0 to 10: 0, no symptom; 1, partial necrosis of the shoot tip; 2, complete necrosis of the shoot tip; 5, complete necrosis on the petiole of terminal leaves; 10, complete necrosis on the main stem. All experiments were conducted twice with triplicates.
For the infiltration assay, a pure compound (50 and 100 μg/ml) was infiltrated into the second true leaves of 4-week-old Chinese pearleaf crab apple seedlings with a needleless syringe (Schaad et al., 1996). After 6-h incubation, E. amylovora TS3128 (1 × 108 cfu/ml of phosphate-buffered saline) was infiltrated into the same position of the leaves treated by the compound, and the inoculated leaves were incubated for 48 h in a humidified box (28°C). To measure bacterial growth, the infiltrated leaves were punched using cork-borers (0.5 cm in diameter), and two disks of leaf were ground in 200 μl of sterilized water using a tissue grinder. Extracted bacterial cells were serially diluted and spotted onto an MGY medium. The plates were incubated for 24 h at 28°C, and then the number of the colony were counted. Data from three biological replicates were collected.
For the quantitative data of this study, analysis was performed at least two times independently with three biological replicates unless indicated. The significance of the data was assessed using the Student t-test, and significant differences (P < 0.05) were indicated with an asterisk.
For the molecular identification of the SFC20201208-M01 isolate, the BenA gene region was amplified from the gDNA of the SFC20201208-M01 isolate. Based on the resulting sequence (446 bp of the amplicon), BLAST results showed that the SFC20201208-M01 isolate belongs to the genus Penicillium and is closed to Penicillium brasilianum CBS 253.55 with 100% of sequence similarity. Phylogenetic analysis also showed that five major clades were reconstructed in the phylogenetic tree, and the SFC20201208-M01 was clustered with P. brasilianum CBS 253.55, P. skrjabinii CBS 439.75, P. onobense CBS 174.81, P. paraherquei CBS 271.83, and P. alagoense URM 8086 (Fig. 1). However, rather than sequence similarity with P. brasilianum CBS 253.55, the SFC20201208-M01 isolate exhibited relatively low sequence similarity with P. skrjabinii CBS 439.75 (96.21%), P. onobense CBS 174.81 (96.41%), P. paraherquei CBS 271.83 (97.54%), and P. alagoense URM 8086 (94.26%) in the cluster. As such, the SFC20201208-M01 isolate was identified as P. brasilianum.
The genus Penicillium is one of the largest groups of fungi, which was composed of over 200 recognized species (Visagie et al., 2014). Penicillium can be grown over a wide range of conditions and environments and produces valuable secondary metabolites (e.g., penicillins, trichodermic acid, and meleagrin) for pharmaceuticals and agrochemicals (Kirk et al., 2008). Several studies demonstrated that Penicillium spp. can improve the phytoremediation efficacy of plants. For example, P. aurantiogriseum 44M-3 not only provided sesame growth-promoting activity but also prevented a plant against Fusarium wilt disease (Dong and Cohen, 2001; Zhao et al., 2021). The polypeptide extract of the P. chrysogenum protected plants against tobacco mosaic virus (Li et al., 2021). Different strains of P. brasilianum have also been known to produce many secondary metabolites, such as cerevisterol and griseofulvin, with various biological activities (Bazioli et al., 2017).
To explore the antibacterial activity of P. brasilianum SFC20201208-M01, the cultures of SFC20201208-M01 grown on various media were investigated for their antibacterial activity against E. amylovora TS3128 (Fig. 2A). When the bacterial cell suspension was treated with culture filtrates derived from four different media (SDB, MEB, PDB, and CDB), the PDB culture filtrate exhibited the strongest antibacterial activity, followed by MEB, SDB, and CDB culture filtrates (Fig. 2B). The MIC and IC50 values of PDB culture filtrate were 1.3% and 0.25%, respectively, whereas MIC values of culture filtrates derived from MEB, SDB, and CDB media were 2.5%, 5%, and 10%, respectively (Fig. 2B and C). According to the antibacterial activity results, the PDB medium was selected for further study to identify the active compound from P. brasilianum SFC20201208-M01.
To identify antibacterial compounds, the culture filtrate derived from the PDB medium was partitioned with EtOAc and n-BuOH sequentially. Of the resulting layers, the EtOAc layer had a promising antibacterial activity against E. amylovora TS3128 with a MIC value of 25 μg/ml, and the n-BuOH layer had a moderate antibacterial activity with a MIC value of 100 μg/ml (Supplementary Table 1). However, no antibacterial activity was observed from the water layer up to a concentration of 200 μg/ml.
Based on the in vitro antibacterial activity results, the active EtOAc layer of the P. brasilianum culture filtrate was further separated by various chromatographic procedures, with the guidance of in vitro antibacterial assays against E. amylovora TS3128 (Fig. 3A). Furthermore, considering that HPLC chromatograms from the P. brasilianum culture filtrate, EtOAc layer, and n-and BuOH layer showed a similar pattern (Fig. 3B), we focused on the EtOAc layer to identify active compounds. Consequently, we obtained four pure compounds from the EtOAc layer and identified two new compounds (3 and 4), along with two known compounds (1 and 2) (Fig. 3C). By comparing their spectroscopic data with previously reported data in the literature (Kang and Kim, 2004; Nguyen et al., 2016), compounds 1 and 2 were determined as known compounds penicillic acid and 5,6-dihydro-6-hydroxypenicillic acid, respectively (Table 1).
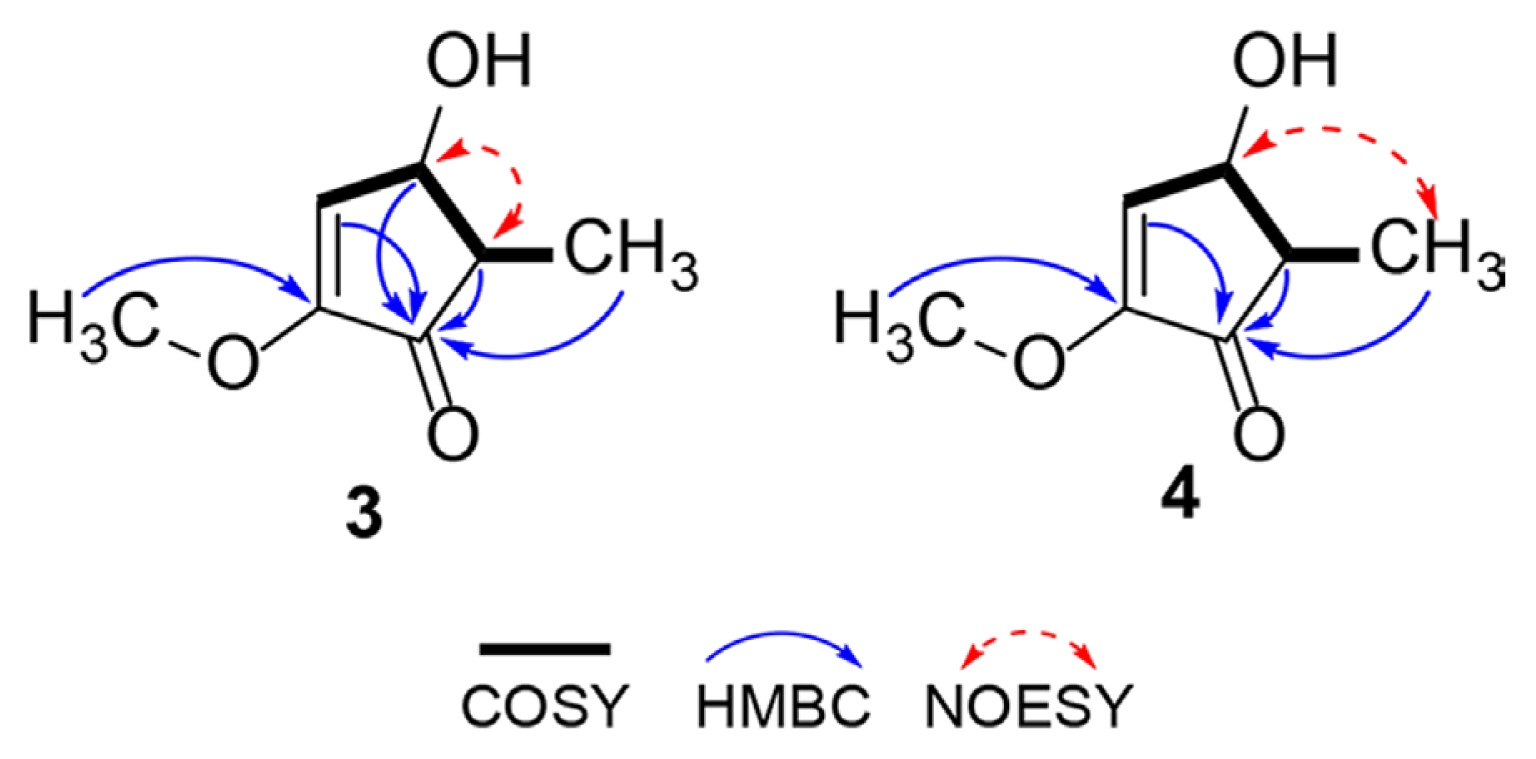
Compound 3 was obtained as a colorless solid. The HRESIMS data showed a quasi-molecular ion at m/z 165.0527 [M + Na]+ (Table 2), indicated the molecular formula C7H10O3 (calculated m/z 165.0528 for C7H10O3Na). The result of zero optical rotation (in MeOH) of 3 indicated that this compound was a racemic form. The 1H, 13C NMR, and heteronuclear single quantum correlation (HSQC) spectra of 3 revealed the presence of a methyl carbon at δC 10.9 (δH 1.11, d, J = 7.6 Hz), a methoxyl carbon at δC 57.7 (δH 3.77, s), an aliphatic methin carbon at δC 45.6 (δH 2.60, qd, J = 7.6, 5.7 Hz), an oxygenated methin carbon at δC 68.1 (δH 4.85, overlapped), an olefinic methin carbon at δC 127.9 (δH 6.44, d, J = 3.2 Hz), a quaternary carbon at δC 158.8, and a ketone carbon at δC 205.8. In the correlated spectroscopy (COSY) analysis of 3, the cross-peak from H-3 to H-6 suggested a part of the structure (Fig. 4). The methoxy carbon was located at C-2 based on the heteronuclear multiple bond correlation (HMBC) analysis from methoxy proton to carbon C-2. Moreover, the correlations from H-3, H-4, H-5, and H-6 to ketone carbon C-1 finished the planar structure of 3, as shown in Fig. 4. Based on the nuclear overhauser effect spectroscopy (NOESY) analysis, the correlation between H-4 and H-5 indicated that both protons were oriented in the same direction, which was confirmed by the large coupling constant (J = 5.7 Hz). Thus, compound 3 was assigned as (4S,5S/4R,5R)-4-hydroxy-2-methoxy-5-methylcyclopent-2-en-1-one and named cis-brasilianone.
Compound 4 was obtained as a colorless solid. The HRESIMS data showed a quasi-molecular ion at m/z 165.0529 [M + Na]+ (Table 2), indicated the molecular formula C7H10O3 (calculated m/z 165.0528 for C7H10O3Na). The result of zero optical rotation (in MeOH) of 4 indicated that this compound was a racemic form. The 1H, 13C NMR, and HSQC spectra of 4 revealed the presence of a methyl carbon at δC 13.3 (δH 1.21, d, J = 7.4 Hz), a methoxyl carbon at δC 57.7 (δH 3.76, d, J = 1.8 Hz), an aliphatic methin carbon at δC 51.1 (δH 2.22, qd, J = 7.4, 1.9 Hz), an oxygenated methin carbon at δC 74.2 (δH 4.39, dd, J = 2.8, 1.9 Hz), an olefinic methin carbon at δC 127.6 (δH 6.39, d, J = 2.8 Hz), a quaternary carbon at δC 158.2, and a ketone carbon at δC 203.9. Detailed COSY and HMBC spectra of 4 were similar to those of 3, suggesting that these compounds shared the same planer structure as shown in Fig. 3C. The different chemical shifts of C-4, C-5, and C-6 of 3 and 4 suggested the different stereochemistry at C-4 and C-5 (Table 2). In the NOESY spectrum, the correlation between H-4 and H-6 indicated the opposite direction of H-4 and H-5, which was confirmed by the small coupling constant (J = 1.9 Hz). Thus, compound 4 was finally assigned as (4S,5R/4R,5S)-4-hydroxy-2-methoxy-5-methylcyclopent-2-en-1-one and named trans-brasilianone.
Penicillic acid (1): Spectroscopic data, see Table 1.
5,6-Dihydro-6-hydroxypenicillic acid (2): Spectroscopic data, see Table 1.
cis-Brasilianone (3): Colorless solid;
[ α ] D 25 0.00
trans-Brasilianone (4): Colorless solid;
[ α ] D 25 0.00
When E. amylovora TS3128 cell suspension was treated with compounds 1-4, penicillic acid (1) exclusively showed antibacterial activity with MIC values of 25 μg/ml (Table 3); compounds 2-4 did not exhibit a bacterial activity against E. amylovora TS3128 up to a concentration of 200 μg/ml. Therefore, penicillic acid was determined as an active compound of P. brasilianum SFC20201208-M01 against E. amylovora.
Since the penicillic acid was firstly isolated from the Penicillium puberulum (Alsberg and Black, 1913), its various biological activities, such as antifungal, antitumor, antidiuretic, and various toxic activities, have been characterized (Ezzat et al., 2007; Kang and Kim, 2004). In terms of antibacterial activity, penicillic acid was found to exert a strong inhibitory effect against Gram-positive and Gram-negative bacteria such as Bacillus cereus, Streptococcus pneumoniae, Pseudomonas aeruginosa, and Staphylococcus aureus (Ezzat et al., 2007). Furthermore, it has been reported that penicillic acid has effects on the morphology of bacteria or acts as an inhibitor of quorum sensing (Auffray et al., 1984; Rasmussen et al., 2005). Nguyen et al. (2016) showed that penicillic acid isolated from Aspergillus persii inhibits the growth of various plant pathogenic bacteria such as Burkholderia glumae, Ralstonia solanacearum, and Xanthomonas arboricola pv. pruni with the MIC values ranging from 12.3 to 111.1 μg/ml, which was consistent with our observations that penicillic acid exhibited antibacterial activity against E. amylovora TS3128 with MIC values of 25 μg/ml.
To determine whether culture filtrate and penicillic acid derived from the P. brasilianum SFC20201208M-01 could prevent fire blight disease, the culture filtrate and penicillic acid were applied onto Chinese pearleaf crab apple seedlings prior to inoculation of E. amylovora TS3128. From the non-treatment control plants, we observed complete necrosis of the terminal buds, petiole, and leaves, whereas there were no symptoms observed from the plant treated with streptomycin at a concentration of 100 μg/ml (Fig. 5). When plants were treated with the culture filtrate, necrosis was limited on terminal buds with a DSI value of 2.0, indicating a control value of 77%, whereas the plants treated with 3-fold diluted culture filtrate exhibited a similar disease development to the non-treatment control plants (Fig. 5A).
When penicillic acid was applied to the plants at a concentration of 125 μg/ml, the penicillic acid sufficiently controlled the development of fire blight with a DSI value of 1.3, which was comparable to the streptomycin-treated control, whereas the non-treatment control plant exhibited a DSI value of 9.6 (Fig. 5B). However, at the concentration of 250 and 500 μg/ml, we observed phytotoxic symptoms 3 days after treatment of penicillic acid. Furthermore, when leaves were infiltrated by E. amylovora TS3128, penicillic acid exhibited a 10- and 20-fold reduction of viable cell numbers at a concentration of 50 and 100 μg/ml, respectively, compared to the non-treatment control (Fig. 5C). When leaves were treated with streptomycin as a positive control, we did not observe any symptoms and viable cells from the leaves (Fig. 5C). Therefore, our results suggest that a low concentration of penicillic acid effectively controls E. amylovora and its disease development.
Currently, a limited number of antibiotics such as streptomycin, oxytetracycline, and oxolinic acid have been registered and used to control E. amylovora. Given the problem of antibiotic resistance, there is no doubt that the development of new antibiotics is urgently needed. In this study, an in vivo control efficacy system using apple seedlings for the fire blight has been established, which can be used to discover and develop novel antibiotics and biological reagents effective in controlling E. amylovora.
Herein, our results showed that P. brasilianum SFC20201208-M01 isolated from a marine environment exhibits a biocontrol potential against fire blight disease caused by E. amylovora. Given that P. brasilianum culture filtrates effectively control fire blight disease, we isolated and identified two new compounds (3 and 4), along with two known compounds (1 and 2). Of these compounds, in vitro and in vivo assays revealed that penicillic acid (1) exhibited promising antibacterial activity against E. amylovora TS3128. Although penicillic acid cannot be used directly as a biocontrol agent because of toxicity, penicillic acid may play an important role as a lead molecule for the development of novel antibiotics for the control of various plant bacterial diseases.
Acknowledgments
This research was supported by the Cooperative Research Program for Agricultural Science and Technology Development (Project PJ01529603), Rural Development Administration, Republic of Korea.
Electronic Supplementary Material
Supplementary materials are available at The Plant Pathology Journal website (http://www.ppjonline.org/).
Fig. 1
Phylogenetic analysis of an isolate SFC20201208-M01 based on the sequence of β-tubulin (BenA) gene.

Fig. 2
Antibacterial activity of an SFC20201208-M01 isolate against Erwinia amylovora TS3128. (A) The cultural phenotype of Penicillium brasilianum SFC20201208-M01 on various media. The upper layer showed cultures grown solid plate, and the lower layer showed cultures grown liquid medium. (B) Antibacterial activity of culture filtrates derived from P. brasilianum SFC20201208-M01 grown in various media against E. amylovora TS3128. SDB, Sabouraud’s dextrose broth; MEB, malt extract broth; PDB, potato dextrose broth; CDB, Czapek-Dox broth medium; NC, normal control. (C) Minimal inhibitory concentration (MIC) and IC50 values of culture filtrate of P. brasilianum SFC20201208-M01 grown on PDB medium at 25°C for 14 days.
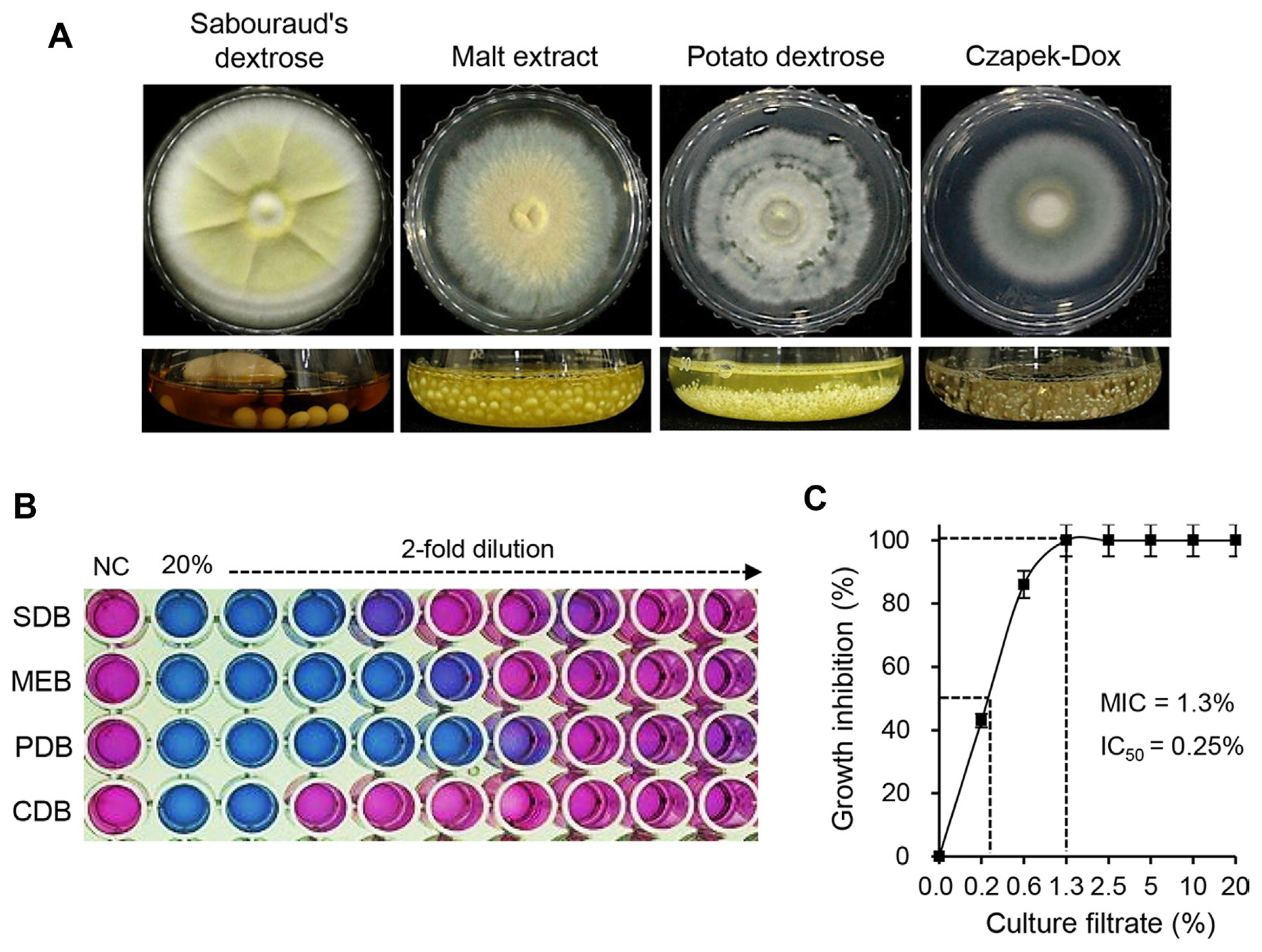
Fig. 3
Isolation and identification of active compounds from Penicillium brasilianum SFC20201208-M01. (A) Isolation scheme of active compounds from P. brasilianum SFC20201208-M01 culture filtrate. (B) High-performance liquid chromatography chromatograms of organic solvent extracts of P. brasilianum SFC20201208-M01 culture. (C) Chemical structures of active compounds identified from P. brasilianum SFC20201208-M01. 1, penicillic acid; 2, 5,6-dihydro-6-hydroxypenicillic acid; 3, cis-brasilianone; 4, trans-brasilianone.
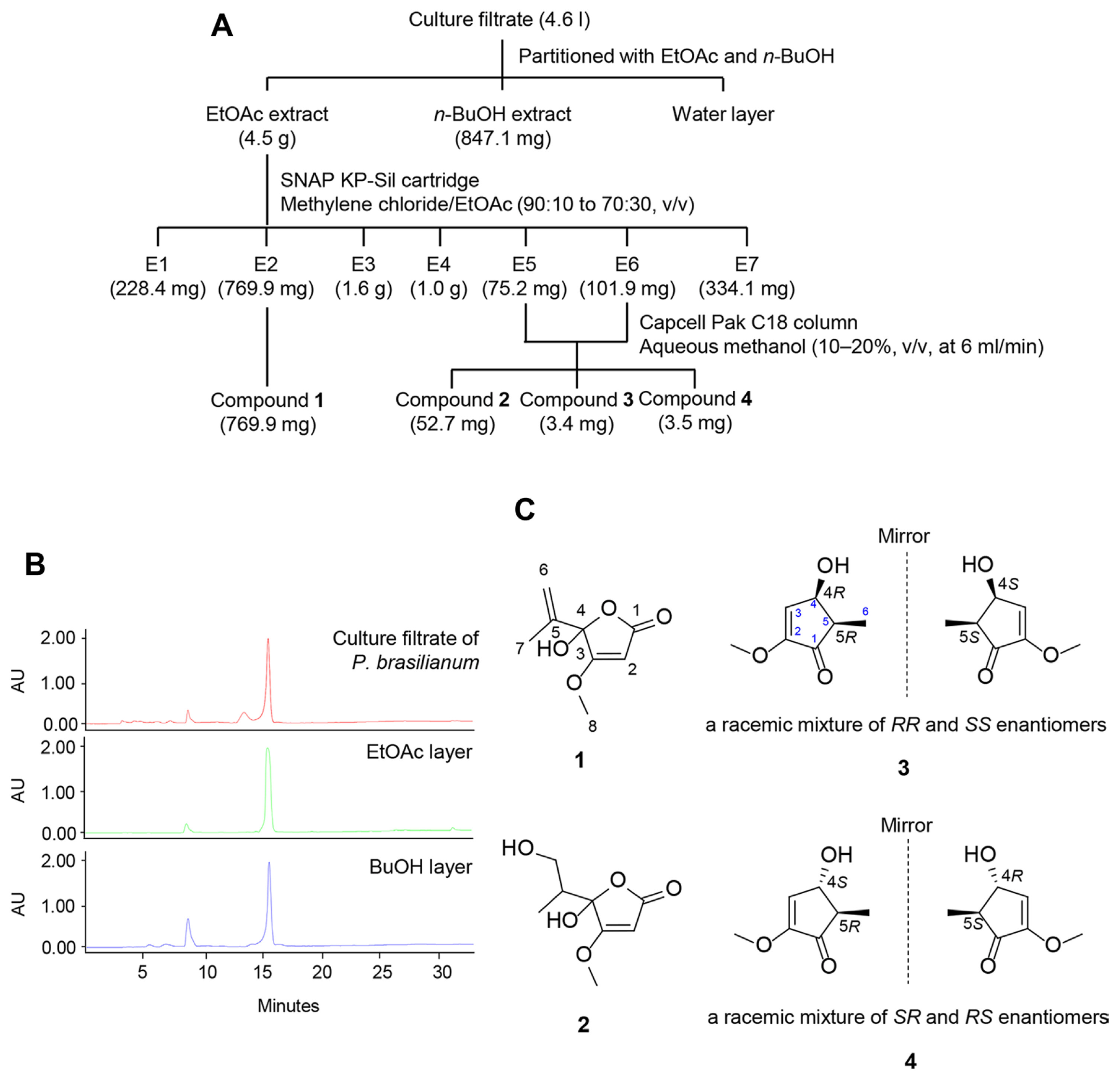
Fig. 5
In vivo antibacterial activity of culture filtrate of Penicillium brasilianum SFC20201208-M01 and penicillic acid. Representatives of Chinese pearleaf crab apple seedlings treated with culture filtrate (A) and penicillic acid (B). Treatments of streptomycin (100 μg/ml) and 1% dimethyl sulfoxide were used as positive and negative controls, respectively. Plants were inoculated with a cell suspension (4 × 108 cfu/ml) of Erwinia amylovora TS3128 at 24 h after the treatment. Photos were taken at 9 dpi. (C) Effect of penicillic acid on bacterial growth on apple leaves. Photos were taken at 48 hpi. The values represent the mean ± standard deviation of two runs with three replicates. Asterisks (*) indicate significant differences at P < 0.05. NC, non-treatment control; DSI, disease severity index; PA, penicillic acid; PT, phytotoxicity; ST, streptomycin.
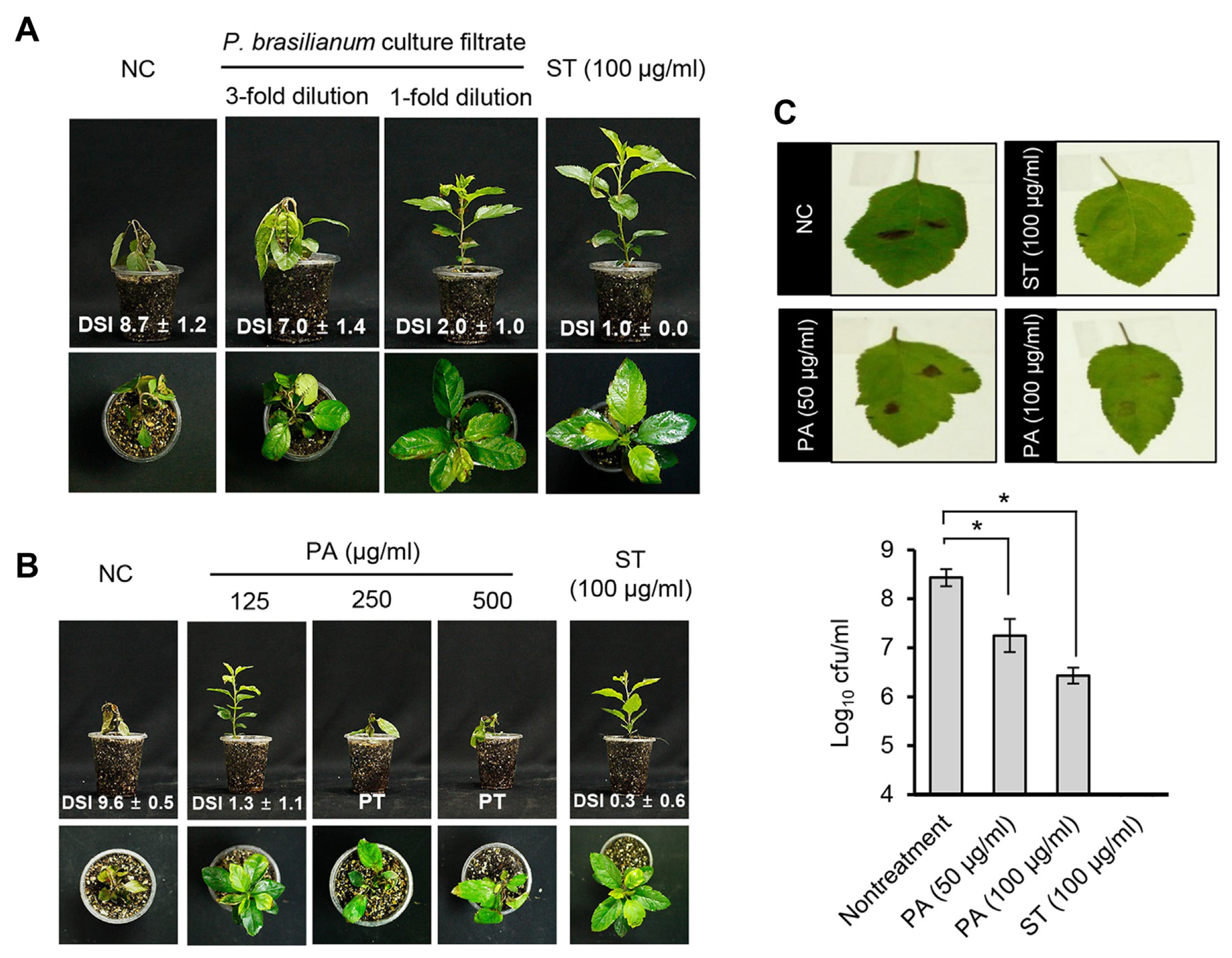
Table 1
1H and 13C NMR data of compounds 1 and 2 (400 MHz in methanol-d4)
Table 2
1H and 13C NMR data of compounds 3 and 4 (400 MHz in methanol-d4)
Table 3
In vitro antibacterial activity of compounds 1-4 against Erwinia amylovora TS3128
References
Aćimović, SG, Zeng, Q, McGhee, GC, Sundin, GW and Wise, JC 2015. Control of fire blight (Erwinia amylovora) on apple trees with trunk-injected plant resistance inducers and antibiotics and assessment of induction of pathogenesis-related protein genes. Front. Plant Sci 6:16.



Alsberg, CL and Black, OF 1913. Contributions to the study of maize deterioration: biochemical and toxicological investigations of Penicillium puberulum and Penicillium stoloniferum
. U.S. Department of Agriculture, Washington, DC, USA. pp. 47.
Auffray, Y, Boutibonnes, P and Lemarinier, S 1984. Filamentous forms of Bacillus thuringiensis (Berliner) formed in the presence of genotoxic mycotoxins. Microbiol. Alim. Nutr 2:59-67.
Bazioli, JM, Amaral, LDS, Fill, TP and Rodrigues-Filho, E 2017. Insights into Penicillium brasilianum secondary metabolism and its biotechnological potential. Molecules 22:858.



Bonaterra, A, Cabrefiga, J, Camps, J and Montesinos, E 2007. Increasing survival and efficacy of a bacterial biocontrol agent of fire blight of rosaceous plants by means of osmoadaptation. FEMS Microbiol. Ecol 61:185-195.


Broggini, GAL, Duffy, B, Holliger, E, Schärer, H-J, Gessler, C and Patocchi, A 2005. Detection of the fire blight biocontrol agent Bacillus subtilis BD170 (Biopro®) in a Swiss apple orchard. Eur. J. Plant Pathol 111:93-100.


Coyier, DL and Covey, RP 1975. Tolerance of Erwinia amylovora to streptomycin sulfate in Oregon and Washington. Plant Dis. Rep 59:849-852.
Dagher, F, Olishevska, S, Philion, V, Zheng, J and Déziel, E 2020. Development of a novel biological control agent targeting the phytopathogen Erwinia amylovora
. Heliyon 6:e05222.



De León Door, AP, Romo Chacón, A and Acosta Muñiz, C 2013. Detection of streptomycin resistance in Erwinia amylovora strains isolated from apple orchards in Chihuahua, Mexico. Eur. J. Plant Pathol 137:223-229.


Dong, H and Cohen, Y 2001. Extracts of killed Penicillium chrysogenum induce resistance against Fusarium wilt of melon. Phytoparasitica 29:421.


Erfani, J, Abdollahi, H, Ebadi, A, Moghaddam, MRF and Arzani, K 2013. Evaluation of fire blight resistance and the related markers in some European and Asian pear cultivars. Seed Plant Improv. J 29:659-672.
Espinel-Ingroff, A, Fothergill, A, Ghannoum, M, Manavathu, E, Ostrosky-Zeichner, L, Pfaller, M, Rinaldi, M, Schell, W and Walsh, T 2005. Quality control and reference guidelines for CLSI broth microdilution susceptibility method (M 38-A document) for amphotericin B, itraconazole, posaconazole, and voriconazole. J. Clin. Microbiol 43:5243-5246.




Ezzat, SM, El-Sayed, EA, Abou El-Hawa, MI and Ismaiel, AA 2007. Morphological and ultrastructural studies for the biological action of penicillic acid on some bacterial species. Res. J. Microbiol 2:303-314.

Glass, NL and Donaldson, GC 1995. Development of primer sets designed for use with the PCR to amplify conserved genes from filamentous ascomycetes. Appl. Environ. Microbiol 61:1323-1330.




Hamed, I, Özogul FÖzogul, Y and Regenstein, JM 2015. Marine bioactive compounds and their health benefits: a review. Compr. Rev. Food Sci. Food Saf 14:446-465.

Hauben, L and Swings, J 2005. Genus XIII. Erwinia. In: Bergey’s manual of systematic bacteriology, Vol. 2. The proteobacteria, 2nd ed. eds. by DJ Brenner, NR Krieg, JT Staley and GM Garrity, pp. 670-679. Springer, New York, NY, USA.
Kang, SW and Kim, SW 2004. New antifungal activity of penicillic acid against Phytophthora species. Biotechnol. Lett 26:695-698.


Kirk, PM, Cannon, PF, Minter, DW and Stalpers, JA 2008. Ainsworth & Bisby’s dictionary of the fungi. 10th ed. CABI Publishing, Wallingford, UK. pp. 771.
Kong, HG, Ham, H, Lee, M-H, Park, DS and Lee, YH 2021. Microbial community dysbiosis and functional gene content changes in apple flowers due to fire blight. Plant Pathol. J 37:404-412.




Li, Y, Jiao, M, Li, Y, Zhong, Y, Li, X, Chen, Z, Chen, S and Wang, J 2021.
Penicillium chrysogenum polypeptide extract protects tobacco plants from tobacco mosaic virus infection through modulation of ABA biosynthesis and callose priming. J. Exp. Bot 72:3526-3539.




Malnoy, M, Martens, S, Norelli, JL, Barny, M-A, Sundin, GW, Smits, THM and Duffy, B 2012. Fire blight: applied genomic insights of the pathogen and host. Annu. Rev. Phytopathol 50:475-494.


McManus, PS, Stockwell, VO, Sundin, GW and Jones, AL 2002. Antibiotic use in plant agriculture. Anuu. Rev. Phytopathol 40:443-465.
Naglot, A, Goswami, S, Rahman, I, Shrimali, DD, Yadav, KK, Gupta, VK, Rabha, AJ, Gogoi, HK and Veer, V 2015. Antagonistic potential of native Trichoderma viride strain against potent tea fungal pathogens in North East India. Plant Pathol. J 31:278-289.



Nguyen, HT, Yu, NH, Jeon, SJ, Lee, HW, Bae, C-H, Yeo, JH, Lee, HB, Kim, I-S, Park, HW and Kim, J-C 2016. Antibacterial activities of penicillic acid isolated from Aspergillus persii against various plant pathogenic bacteria. Lett. Appl. Microbiol 62:488-493.



Norelli, JL, Jones, AL and Aldwinckle, HS 2003. Fire blight management in the twenty-first century: using new technologies that enhance host resistance in apple. Plant Dis 87:756-765.


Oh, M, Son, H, Choi, GJ, Lee, C, Kim, J-C, Kim, H and Lee, Y-W 2016. Transcription factor ART1 mediates starch hydrolysis and mycotoxin production in Fusarium graminearum and F. verticillioides
. Mol. Plant Pathol 17:755-768.


Park, DH, Yu, J-G, Oh, E-J, Han, K-S, Yea, MC, Lee, SJ, Myung, I-S, Shim, HS and Oh, C-S 2016. First report of fire blight disease on Asian pear caused by Erwinia amylovora in Korea. Plant Dis 100:1946.

Pusey, PL, Stockwell, VO, Reardon, CL, Smits, THM and Duffy, B 2011. Antibiosis activity of Pantoea agglomerans biocontrol strain E325 against Erwinia amylovora on apple flower stigmas. Phytopathology 101:1234-1241.


Rasmussen, TB, Skindersoe, ME, Bjarnsholt, T, Phipps, RK, Christensen, KB, Jensen, PO, Andersen, JB, Koch, B, Larsen, TO, Hentzer, M, Eberl, L, Hoiby, N and Givskov, M 2005. Identity and effects of quorum-sensing inhibitors produced by Penicillium species. Microbiology 151:1325-1340.


Roselló, G, Bonaterra, A, Francés, J, Montesinos, L, Badosa, E and Montesinos, E 2013. Biological control of fire blight of apple and pear with antagonistic Lactobacillus plantarum
. Eur. J. Plant Pathol 137:621-633.


Russo, NL, Burr, TJ, Breth, DI and Aldwinckle, HS 2008. Isolation of streptomycin-resistant isolates of Erwinia amylovora in New York. Plant Dis 92:714-718.


Saitou, N and Nei, M 1987. The neighbor-joining method: a new method for reconstructing phylogenetic trees. Mol. Biol. Evol 4:406-425.

Schaad, NW, Wang, ZK, Di, M, McBeath, J, Peterson, GL and Bonde, MR 1996. An improved infiltration technique to test the pathogenicity of Xanthomonas oryzae pv. oryzae in rice seedlings. Seed Sci. Technol 24:449-456.
Schroth, MN, Thomson, SV, Hildebrand, DC and Moller, WJ 1974. Epidemiology and control of fire blight. Anuu. Rev. Phytopathol 12:389-412.

Sholberg, PL, Bedford, KE, Haag, P and Randal, P 2001. Survey of Erwinia amylovora isolates from British Columbia for resistance to bactericides and virulence on apple. Can. J. Plant Pathol 23:60-67.

Sobiczewski, P, Peil, A, Mikiciński, A, Richter, K, Lewandowski, M, Żurawicz, E and Kellerhals, M 2015. Susceptibility of apple genotypes from European genetic resources to fire blight (Erwinia amylovora). Eur. J. Plant Pathol 141:51-62.


Thomson, SV, Gouk, SC, Vanneste, JL, Hale, CN and Clark, RG 1993. The presence of streptomycin resistant isolates of Erwinia amylovora in New Zealand. Acta. Hortic 338:223-230.
Vanneste, JL 2000. Fire blight: the disease and its causative agent. Erwinia amylovora. CABI publishing, Wallingford, UK. pp. 370.
Visagie, CM, Houbraken, J, Frisvad, JC, Hong, S-B, Klaassen, CHW, Perrone, G, Seifert, KA, Varga, J, Yaguchi, T and Samson, RA 2014. Identification and nomenclature of the genusPenicillium
. Stud. Mycol 78:343-371.



Vrancken, K, Holtappels, M, Schoofs, H, Deckers, T and Valcke, R 2013. Pathogenicity and infection strategies of the fire blight pathogen Erwinia amylovora in Rosaceae: state of the art. Microbiology 159:823-832.





 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Full text via PMC
Full text via PMC Download Citation
Download Citation Supplement
Supplement Print
Print




