DNA Microarray and Gene Ontology Enrichment Analysis Reveals That a Mutation in opsX Affects Virulence and Chemotaxis in Xanthomonas oryzae pv. oryzae
Article information
Abstract
Xanthomonas oryzae pv. oryzae (Xoo) causes bacterial leaf blight (BLB) in rice (Oryza sativa L.). In this study, we investigated the effect of a mutation in opsX (XOO1056), which encodes a saccharide biosynthesis regulatory protein, on the virulence and bacterial chemotaxis of Xoo. We performed DNA microarray analysis, which showed that 63 of 2,678 genes, including genes related to bacterial motility (flagellar and chemotaxis proteins) were significantly downregulated (<−2 log2 fold changes) by the mutation in opsX. Indeed, motility assays showed that the mutant strain was nonmotile on semisolid agar swarm plates. In addition, a mutant strain (opsX::Tn5) showed decreased virulence against the susceptible rice cultivar, IR24. Quantitative real-time RT-PCR reaction was performed to confirm the expression levels of these genes, including those related to flagella and chemotaxis, in the opsX mutant. Our findings revealed that mutation of opsX affects both virulence and bacterial motility. These results will help to improve our understanding of Xoo and provide insight into Xoo-rice interactions.
Introduction
Bacterial leaf blight (BLB), caused by Xanthomonas oryzae pv. oryzae (Xoo), is a major disease affecting rice grown in Asian countries. The opsX gene, which encodes a saccharide biosynthesis regulatory protein required for the assembly of lipopolysaccharide (LPS), has been cloned from Xanthomonas campestris pv. citrumelo (Kingsley et al., 1993). LPS from several plant pathogens have been shown to activate a number of defense-related responses in plants. LPS from X. campestris can induce generation of transcripts for a defense-related β-1,3-glucanase in turnip (Newman et al., 1995), purified LPS from Pseudomonas syringae pv. syringae is a weak elicitor of phytoalexins in soybean (Barton-Willis et al., 1984), and LPS from Ralstonia solanacearum can induce the synthesis of new polypeptides in tobacco (Leach et al., 1983). Mutation of opsX resulted in a loss of virulence on the citrus host, marked by rapid cell death, although the hypersensitive response (HR), host range, and virulence to beans remained unaffected (Kingsley et al., 1993). Although it is tempting to conclude that LPS is responsible for protection from plant inhibitors, mutations in opsX also have pleiotropic effects on growth rate, capsular slime, and extracellular polysaccharide (EPS). Indeed, Kingsley et al. (1993) reported that opsX of X. oryzae pv. citrumelo affects biosynthesis of LPS and EPS. In the M28 mutant (ΔopsX), derived from wild-type X. oryzae pv. citrumelo strain 3048, loss of LPS decreased EPS production to one-third that of the wild type. Motility is an important virulence factor in many pathogenic species and in some cases is inversely regulated with the expression of other virulence traits (Ottemann and Miller, 1997). Although the chemotactic properties of xanthomonads have not been extensively studied, Feng and Kuo (1975) reported that the rice pathogen Xoo showed chemotaxis toward a synthetic medium and water exudates of susceptible rice plants, whereas chemotaxis toward hydathode exudates of resistant plants was not observed. In addition, X. campestris pv. campestris is nonmotile and nonflagellated in the xylem fluid of infected cabbage leaves, which suggests that bacterial movement in the xylem is due to diffusion rather than to motility (Kamoun and Kado, 1990). This suggests that chemotaxis may play a role before the bacteria penetrate the host leaf. However, the role of chemotaxis in bacterial movement inside the plant remains unknown (Kamoun and Kado, 1990).
This study investigated the effect of opsX mutation on virulence and xanthan and LPS production in Xoo, as well as on the expression of other genes that are known to play roles in chemotaxis.
Materials and Methods
Bacterial strains and culture conditions
The Xoo KACC10859 strain was obtained from the Korean Agricultural Culture Collection (KACC) in Jeonju, Korea, and was cultured on yeast dextrose carbonate (YDC) medium (2.0% D-glucose, 2.0% CaCO3, 1.0% yeast extract, and 1.5% agar) or in nutrient broth (3% beef extract, 5% peptone, and 1.5% agar, Difco; DB, Seoul, Korea) at 28ºC until 2.0 OD600. Xoo strains were washed twice and immediately transferred into XOM2 medium (0.18% xylose sugar, 670 mM D-, L-methionine, 10 mM sodium L(+)-glutamate, 14.7 mM KH2PO4, 40 mM MnSO4, 240 mM Fe (III) ethylenediaminetetraacetic acid (EDTA), and 5 mM MgCl2, pH 6.5).
Transposon mutagenesis and molecular analysis of the opsX mutant
Insertional mutagenesis was performed using transposome™ (20 ng/ml; Epicentre, Seoul, Korea) and the insertion site was analyzed by polymerase chain reaction (PCR) according to a previously described method (Park et al., 2007). Sequencing and subsequent data analysis were performed using an ABI Prism 3100 automatic DNA sequencer (Thermo Scientific, Seoul, Korea) and sequences were analyzed for similarities using the BLASTN and BLASTX algorithms available at the National Center for Biotechnology Information (NCBI). The opsX mutant was analyzed by PCR with the primers OPSXF (5′-ATGGCTGTAACGCCCGCATC-3′) and OPSXR (5′-TCAGCGCGGGTGGTCGGCGC-3′), corresponding to the opsX (XOO1056) open reading frame (ORF). The PCR was carried out in a PTC-225 thermocycler (Bio-Rad, Seoul, Korea) with the following conditions: initial denaturation at 95ºC for 5 minutes followed by 30 cycles at 95ºC for 30 seconds, 65ºC for 30 seconds, and 72ºC for 2 minutes, and a final extension for 5 minutes at 72ºC. For Southern hybridization, genomic DNA was digested with EcoRI (Toyobo, Seoul, Korea), electrophoresed, and transferred to a Hybond-N+ membrane (GE Healthcare, Seoul, Korea). The kanamycin gene in the transposon was amplified by PCR with the primers KANF (5′-CAATCAGGTGCGACAATC-3′) and KANR (5′-TCACCGAGGCAGTTCCAT-3′), and labeled as a probe with [α-32P] dCTP (Ladderman Labeling Kit; TaKaRa, Seoul, Korea). Prehybridization and hybridization were conducted as described by Sambrook and Russell (2001). For complementation, a DNA fragment containing opsX was amplified from wild-type genomic DNA by PCR using the primers OPSXFC (5′-CTGCAGGAATTCGGCAAAACTGCG CG-3′; PstI restriction site is underlined) and OPSXRC (5′-CTAGCGCGATAGCTTTGAGC-3′), and the PCR product was cloned into the pGEM-T easy vector (Promega, Seoul, Korea). The resulting plasmid was digested with PstI, ligated into the corresponding sites of the broad host-range vector pHMI, and introduced into the opsX mutant strain to obtain the complemented strain (MT/C).
Oligonucleotide and target preparation
DNA microarrays with 50-bp oligo spots representing 3382 ORFs, approximately 3/4 of the Xoo KACC10331 genome, were synthesized and spotted on addressable electrodes of CustomArray™ 12K microarrays. Microarray analysis was performed according to the CustomArray™ 12K microarray protocol provided by CombiMatrix (CombiMatrix Corp.; http://www.combimatrix.com).
RNA was isolated from a stationary phase culture (OD600 = 0.8) using an RNeasy mini kit in accordance with the manufacturer’s instructions (Qiagen, Seoul, Korea). Total RNA was eluted with 50 μl of RNase-free water and quantified using an ultraviolet spectrophotometer (ND-1000). Each sample was run on a 1.2% agarose gel to verify the purity and integrity of the RNA. cDNA was synthesized using the MessageAmp™ II-Bacteria Kit (Thermo Scientific). Polyadenylation of the bacterial RNA was performed using Escherichia coli poly(A) polymerase and tailed RNA reverse-transcribed in a reaction mixture primed with an oligo (dT) primer and ArrayScript™ (Thermo Scientific) reverse transcriptase. Second-strand cDNA was synthesized and purified for in vitro transcription in a reaction mixture containing biotinmodified UTP and T7 RNA polymerase.
Microarray hybridization
Hybridization was performed using 5 μg of labeled target sample per CustomArray™ 12K microarray for 16 hours at 45ºC with gentle rotation. The arrays were then washed with: (i) 6 × SSPE, 0.05% Tween-20 for 5 minutes, (ii) 3 × SSPE, 0.05% Tween-20 for 1 minute, (iii) 0.5 × SSPE, 0.05% Tween-20 for 1 minute, and (iv) 2 × phosphate-buffered saline, 0.1% Tween-20 for 1 minute.
Data acquisition and analysis
Hybridized microarrays were scanned (photomultiplier tube, 500–700; pixel size, 5; focus position, 130) using a GenePix 4000B microarray scanner (Axon Instruments, Union City, CA, USA). After data extraction (https://webapps.combimatrix.com), the background was calculated for individual samples using factory-built controls with low intensities (lowest intensity, 5–30%) and the median signal intensities were calculated for subtraction. Microarray data of individual samples were normalized by global normalization using probes with signal values greater than zero, < 60,000 (the saturation value), and greater than the lowest 5% of the signal value of each sample. In total, 3,382 probes were used for the final analysis. The local-pooled-error (LPE) test (http://bioinformatics.oxfordjournals.org/cgi/reprint/19/15/1945) and fold-change analysis were applied to determine differentially expressed sets of genes using the Avadis Prophetic software version 3.3 (Strand Genomics Ltd., Bangalore, India). For further analysis of the upregulated and downregulated genes, gene ontology (GO) term annotation (biological process, molecular function, and cellular component) and enrichment analysis were performed using the Blast2GO software with default parameters (Conesa et al., 2005).
Virulence assay
Leaves of 50-day-old susceptible rice (cultivar IR24) were inoculated with the pathogen by clipping the leaf tips with sterile scissors dipped in a saturated culture (109 cells/ml). The rice plants were then grown in a greenhouse at 25–30ºC with a relative humidity of 60%. Leaves clipped with scissors dipped in distilled water were used as the control. Lesion length was measured 21 days after inoculation. The virulence assay was performed in triplicate for both wild-type and mutant strains, and means and standard deviations (SDs) of lesion lengths were calculated for each experiment.
Determination of EPS and LPS production
The amount of xanthan in the supernatant was determined by a modified version of the phenol-sulfuric acid method (DuBois et al., 1956). Culture supernatant without bacterial cells (filtered through a 0.45-mm filter; Sigma- Aldrich, Yongin, Korea) was tested to estimate the amount of xanthan. Culture supernatant (0.3 ml) was mixed with 1.65 ml of distilled water and 0.05 ml of 80% (v/v) phenol. Then, 5 ml of 100% sulfuric acid was added to the phenol mixture, suspended, and placed at room temperature for 30 minutes. The concentration of xanthan was spectrophotometrically determined by measuring the absorbance at 490 nm. LPS was extracted using the hot phenol-water method described by Westphal and Jann (1965), subjected to 12% sodium dodecyl sulphate polyacrylamide gel electrophoresis (SDS-PAGE), and visualized via silver staining (Tsai and Frasch, 1982). Nucleic acids were removed by incubation with DNase (Qiagen) and RNase (Qiagen) for 1 hour at 37ºC. Proteins were removed by incubation with protease (Qiagen) for 1 hour at 37ºC.
Quantitative real-time RT-PCR assay
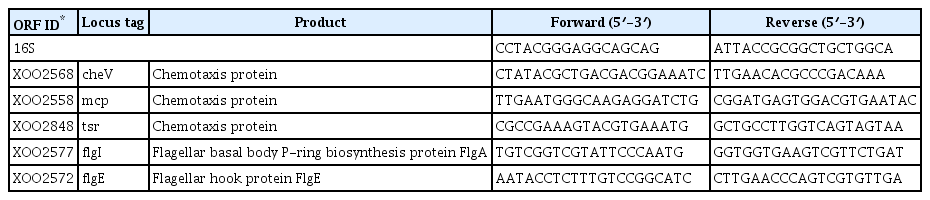
An independent set of cell cultures and total RNA of the two Xoo strains were prepared following the same protocol as for the microarray analysis. The sequence of each gene was obtained from the Xoo KACC10331 database (http://www.ncbi.nlm.nih.gov), and used for primer design by IDT (Integrated DNA Technologies, https://eu.idtdna.com/Primerquest/Home/Index). RNA samples from three independent replicates were treated with DNase before cDNA synthesis. Quantitative real-time PCR analysis was performed using a RotorGene 6000 (Qiagen) in 25-μl reactions containing 12.5 μl of 2X SensiFAST SYBR No-ROX kit (Bioline, Sydney, Australia), 10 pmol of each primer (Table 1), and 25 ng of cDNA template. The following reaction conditions were used: 3 minutes at 95ºC followed by 40 cycles of 95ºC for 5 seconds, 60ºC for 10 seconds, and 72ºC for 15 seconds.
Swarming and growth rate assays
Bacterial motility was assayed using a previously described method (Kim et al., 2015). Cultures were grown in nutrient broth (NB) medium for 24 hours, washed 3 times with 1 ml of PME buffer (10 mM potassium phosphate [pH 7.2], 1 mM MgCl2, and 0.1 mM EDTA), and then stabbed into swarm agar plates (0.7% NaCl, 0.115% K2HPO4, 0.02% KH2PO4, 0.02% KCl, 0.01% yeast extract, and 0.3% agar). For the growth rate assay, fifty microliters of each culture (OD600 = 1), prepared from 5 ml of seed culture in NB medium, was inoculated in 5 ml of swarming medium without agar and incubated for 72 hours at 28ºC in a shaking incubator. The ODs of each strain were measured using a GENESYS™ 20 Visible Spectrophotometer (Thermo Scientific). The averages and SDs of swarming and growth rates were calculated for each repetition of the experiment.
Results
Molecular characterization of the opsX mutant
Sequencing analysis revealed that the transposon was inserted at nucleotide 838 of opsX (Fig. 1A). This gene harbors a 1,050-bp ORF encoding a 349-amino acid saccharide biosynthesis regulatory protein. Transposon insertion into opsX was confirmed by PCR amplification of a 2,271-bp fragment from the mutant using the OPSXF and OPSXR primers (Fig. 1B). In the Southern hybridization analysis, a single positive signal (2,513-bp) was detected in the mutant by using the PCR product of the kanamycin-resistant gene as a probe (Fig. 1C). These results indicate that a single transposon was inserted into opsX.
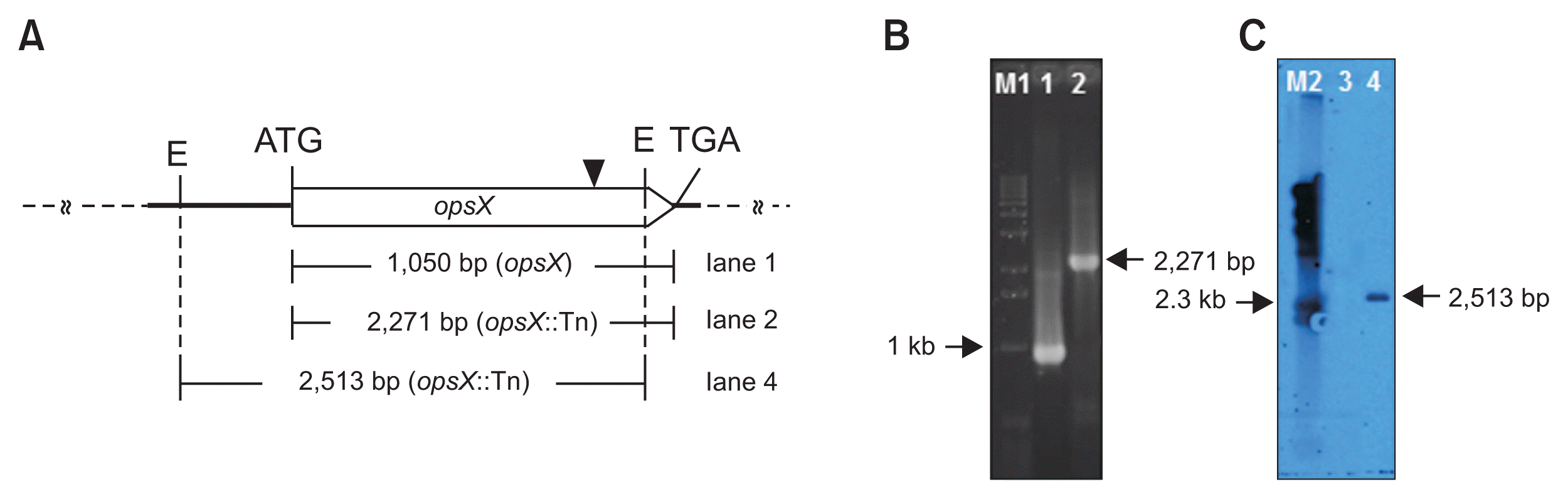
Genetic mapping and molecular analysis of the opsX mutant strain. (A) The solid triangle indicates the site of the Tn5 transposon insertion. Restriction sites for EcoRI (E) are also indicated. (B, C) PCR and Southern hybridization analysis for confirmation of transposon insertion in opsX: M1, 1-kb ladder; M2, lambda DNA/HindIII; lane 1, wild type; lanes 2 and 4, opsX mutant. The arrowhead indicates a single positive signal from the transposon (2,513-bp).
Expression profiles and quantitative real-time RT-PCR
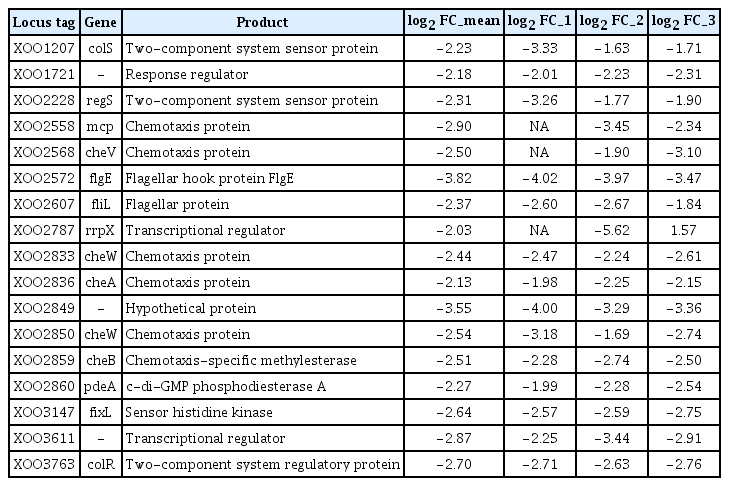
The Xoo DNA-chip was used to compare the expression profile of the opsX mutant with that of the wild-type strain. The resultant expression profile was refined using a false discovery rate of 5% (P < 0.05; LPE test) and a log2 fold change (FC) minimum of ± 2; 259 genes were found to be upregulated or downregulated in the opsX mutant (Table S1). The transcripts of these upregulated and downregulated genes were categorized according to their annotated function with respect to biological processes, molecular functions, and cellular components, on the basis of blast and GO term annotation using the Blast2GO software (Conesa et al., 2005). The biological processes mediated by these genes were primarily associated with metabolic processes, cellular processes, single organism processes, localization, signaling, and others (Fig. 2). Only upregulated genes were found to belong to the multi-organism process and immune system process categories. In contrast, only downregulated genes were found to belong to the developmental process category. Molecular functions were primarily related to catalytic and binding activity. Only upregulated genes belonged to the structural molecule activity category. The cellular component categories included cell, membrane, macromolecular complex, organelle, and extracellular region. Only upregulated genes belonged to the extracellular region category. Most of these categories, such as locomotion, molecular transducer activity, membrane, and organelle, contained more downregulated genes than upregulated genes, whereas the cellular process, metabolic process, single-organism process, localization, catalytic activity, binding, and cell categories contained more upregulated than downregulated genes (Fig. 2, Table S2). In GO term enrichment analysis, four remarkably enriched GO terms, cell motility (GO:0048870), signal transducer activity (GO:0004871), signal transduction (GO:0007165), and kinase activity (GO:0016301), were identified in the downregulated genes (Table 2). Among them, six chemotaxis-related genes (XOO2558, XOO2568, XOO2833, XOO2836, XOO2850, and XOO2859) and two flagellar-related genes (XOO2572 and XOO2607) were downregulated by opsX mutation. The log2 FC levels of these eight genes ranged from −2.13 to −3.82. Among the eight genes, flgE (XOO2572) and cheW (XOO2850) were most strongly downregulated, by −3.82 FC and −2.54 FC, respectively, by mutation of opsX (Table 3). Consistent with the microarray analysis, five genes (cheV, XOO2568; mcp, XOO2558; tsr, XOO2848; flgI, XOO2577; and flgE, XOO2572) involved in bacterial motility showed relatively lower expression in the opsX mutant than in the wild-type strain (Fig. 3).
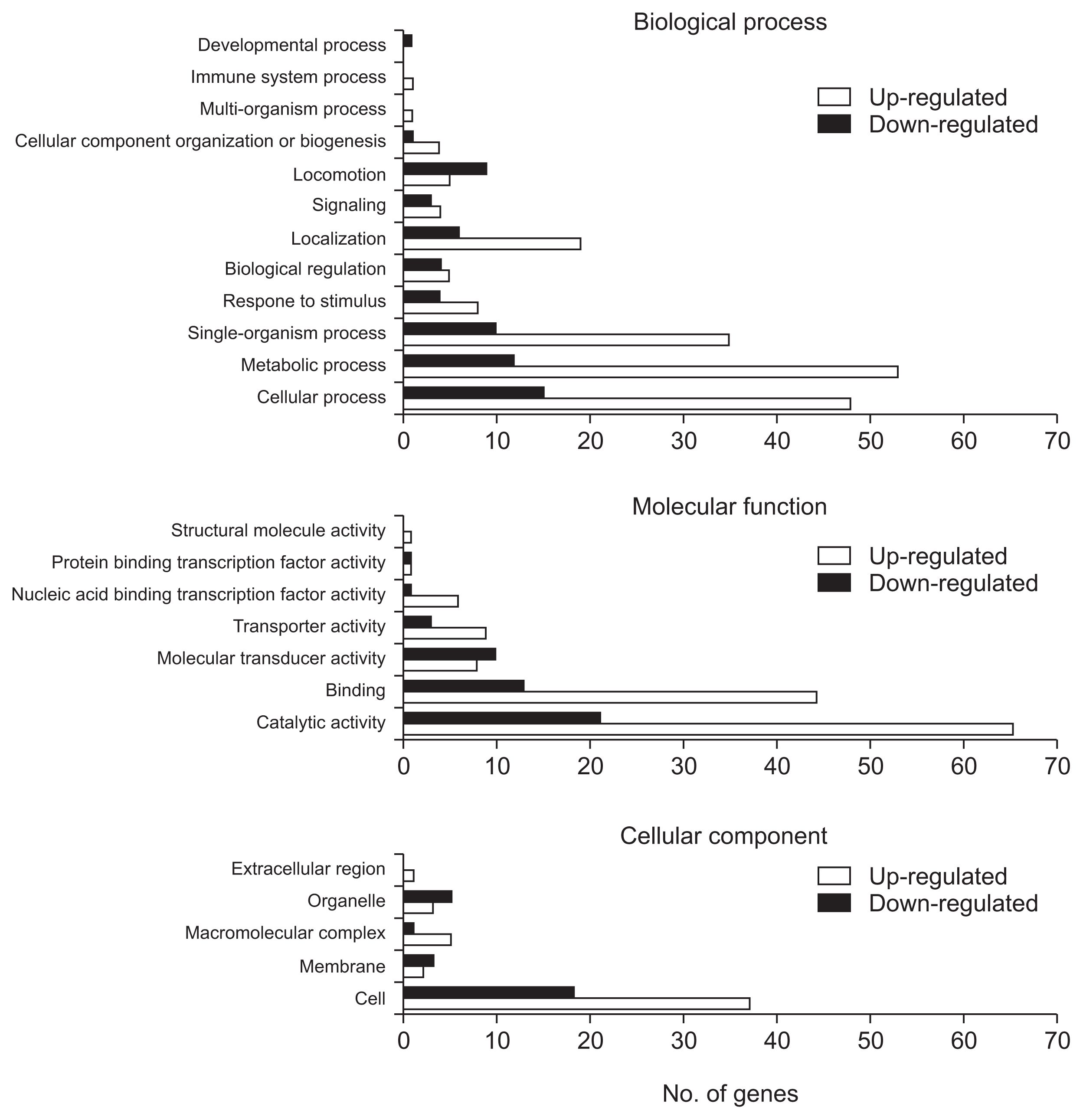
Functional categorization of up- and down-regulated genes in the Xanthomonas oryzae pv. oryzae opsX mutant based on GO annotation.
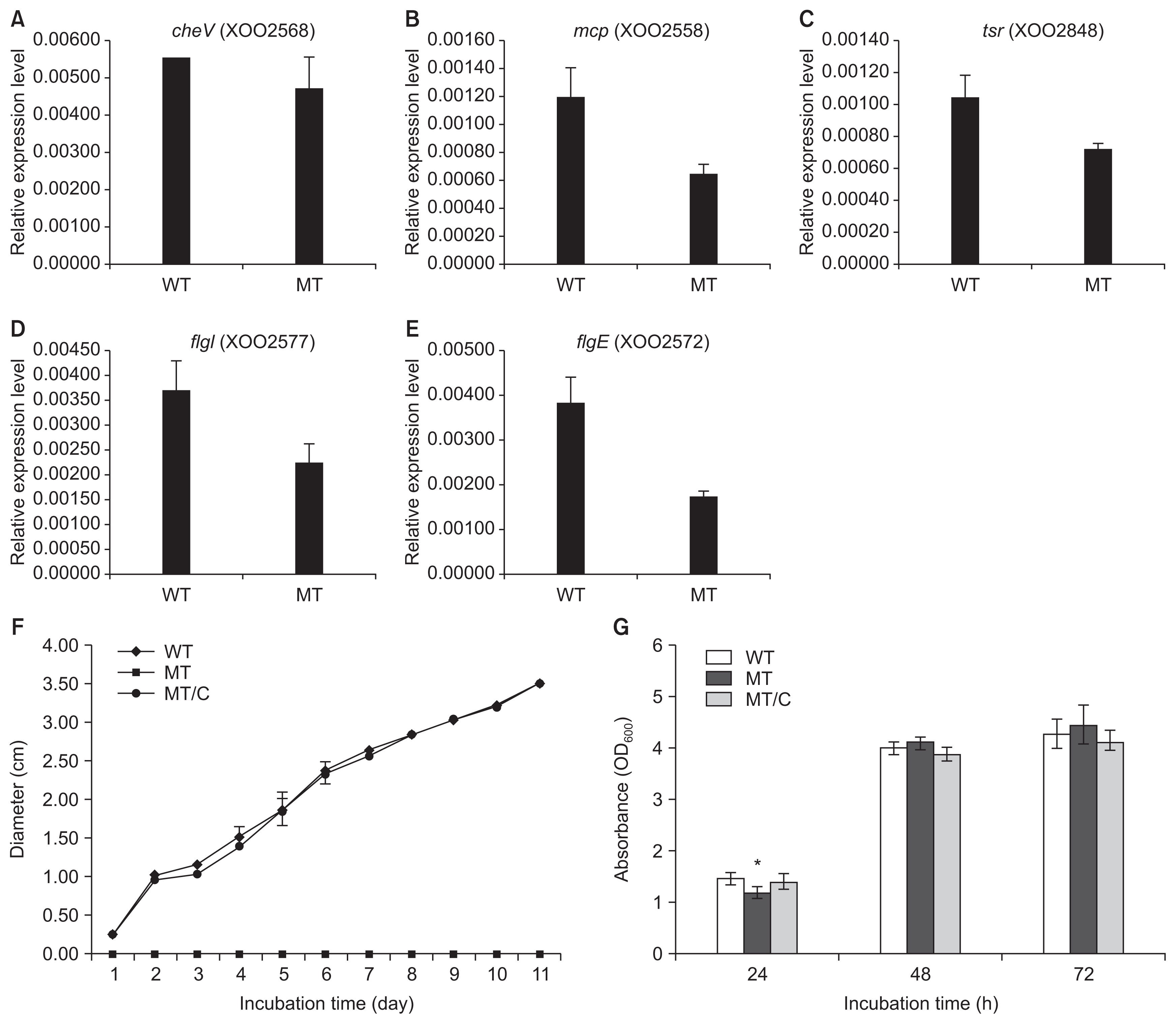
The expression patterns of genes associated with bacterial chemotaxis (cheV, A; mcp, B; tsr, C; flgI, D; and flgE, E) and effect of opsX mutation on swarming (F) and growth rate (G) of Xanthomonas oryzae pv. oryzae wild-type and opsX mutant strains. The gene expression levels (arbitrary units) were normalized using 16S RNA as an internal reference. Gene expression levels were quantified by real-time RT-PCR. WT, wild-type strain; MT, opsX mutant strain; MT/C, complemented strains. The asterisk represents statistically significant differences relative to the wild-type strain (paired, two-tailed Student’s t-test, *P < 0.05).
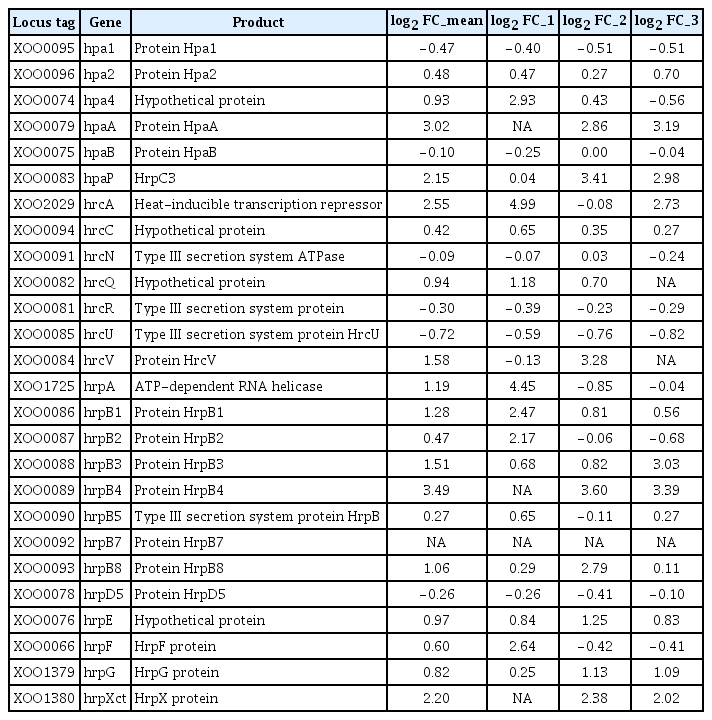
The microarray analysis showed that mutation of opsX in Xoo affected expression of other genes crucial for virulence, including HR and pathogenicity (hrp) genes. According to the genome sequence of Xoo KACC10331, the hrp gene cluster is composed of nine hrp (hrpF, hrpE, hrpD6, hrpD5, hrpB1, hrpB2, hrpB4, hrpB5, and hrpB7), nine hrc (hrp conserved; hrcS, hrcR, hrcQ, hrcV, hrcU, hrcJ [hrpB3], hrcN, hrcT [hrpB8], and hrcC) and eight hpa (hrp-associated; hpaF, hpaB, hpaA, hpaP, hpa1, hpa2, hpa3, and hpa4) genes (Cho et al., 2008; Lee et al., 2005). Interestingly, six hrp genes were slightly downregulated, including hrcU (−0.72 log2 FC), hpa1 (−0.47 log2 FC), hrcR (−0.30 log2 FC), hrpD5 (−0.26 log2 FC), hpaB (−0.10 log2 FC), and hrcN (−0.09 log2 FC) (Table 4), which are known to be crucial for the HR and virulence in Xoo (Cho et al., 2008).
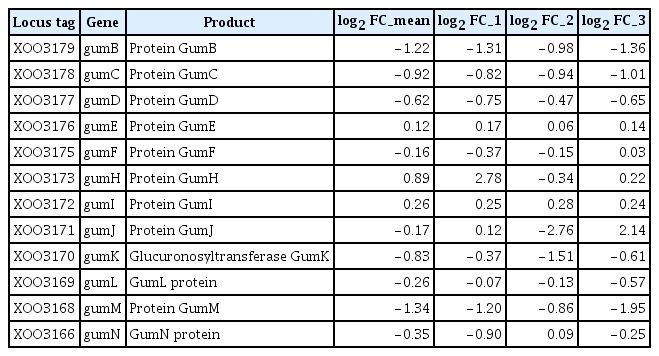
Xanthomonas spp. produce a characteristic EPS, xanthan, a polymer composed of repeating pentasaccharide units that protects bacteria from environmental stresses, including dehydration and toxic compounds (Denny, 1995). The production of xanthan is directed by the gum gene cluster, which consists of 12 genes (gumB to gumM) and is highly conserved among Xanthomonas spp. (Katzen et al., 1998; Vojnov et al., 1998). In addition, the complete genome sequence of Xoo KACC10331 revealed a cluster of ORFs in the genome that consisted of a tandem array of 14 ORFs, gumB, -C, -D, -E, -F, -G, -H, -I, -J, -K, -L, -M, XOO3167, and -N, similar to the gum operon of X. campestris (Lee et al., 2005; Yoon and Cho, 2007). In this study, 9 gum genes (XOO3166, XOO3168, XOO3169, XOO3170, XOO3171, XOO3175, XOO3177, XOO3178, and XOO3179) were downregulated by opsX mutation. The log2 FC levels of the 9 genes ranged from −0.16 to −1.34 (Table 5). However, no GO term enrichment was observed for these downregulated gum genes (Table 3).
Virulence assay, xanthan, and LPS production
In the virulence assay, the opsX mutant showed a severe decrease in virulence against susceptible rice. Twenty-one days after inoculation, the average length of lesions caused by the opsX mutant was 1.67 cm, whereas that of lesions caused by the wild-type strain was 17.67 cm (Fig. 4). In the complementation test, lesions caused by the MT/C strain averaged 15.33 cm, similar to those caused by the wild-type strain (Fig. 4).
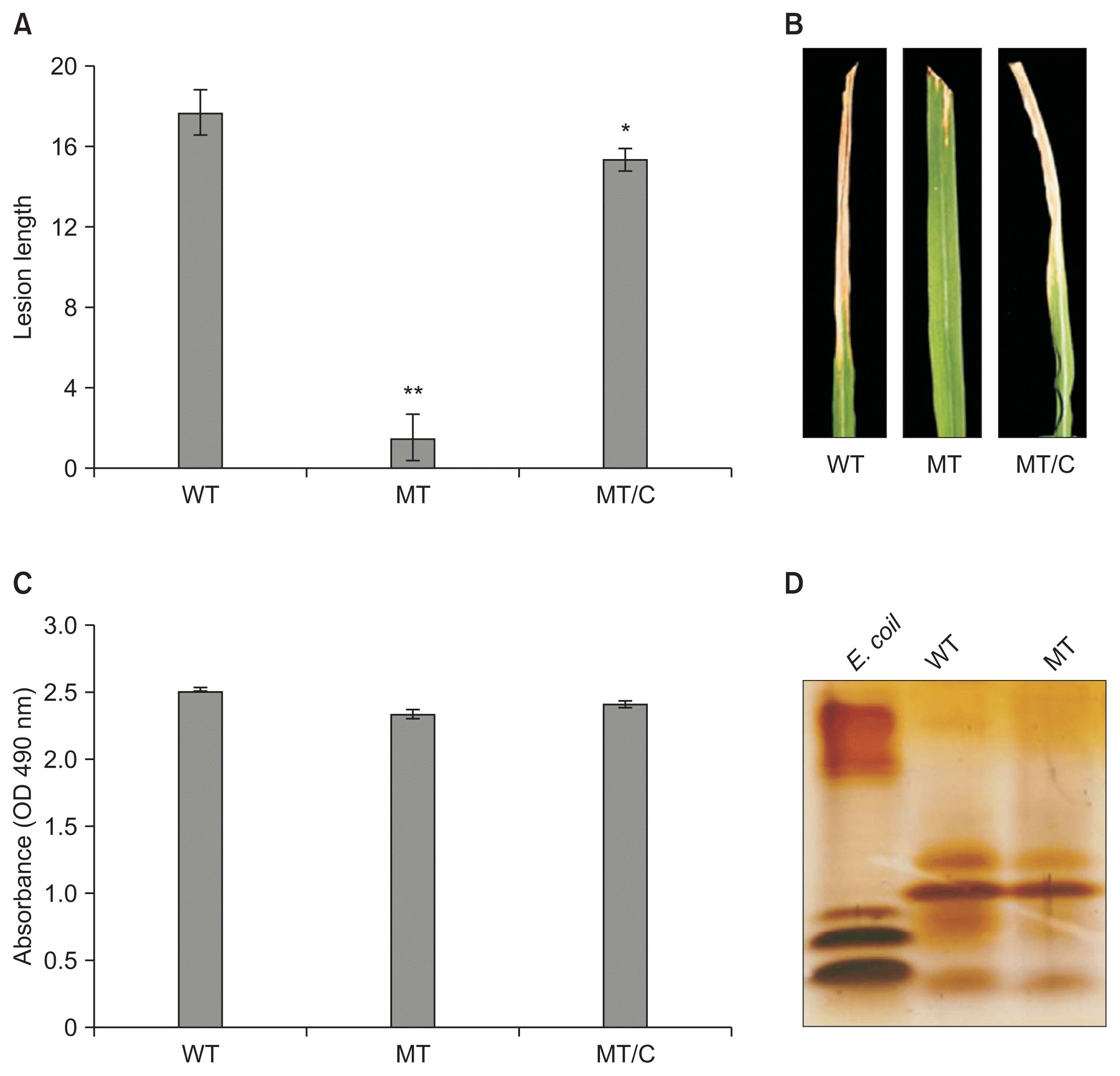
Virulence (A, B), xanthan (C), and lipopolysaccharide (LPS) production assays (D) of wild-type (WT), mutant (MT), and complemented strains (MT/C). Assays were performed in triplicate, and the means and standard deviation for lesion length and xanthan production were calculated for each experiment. The ladder-like structure of LPS from Escherichia coli was used as a comparative sample. The asterisks represent statistically significant differences relative to the WT strain (paired, two-tailed Student’s t-test, *P < 0.05, **P < 0.01).
Although transcriptome analysis revealed that genes related to xanthan biosynthesis were downregulated in the opsX mutant strain, the wild-type and opsX mutant strains did not differ significantly in xanthan production. Mutation of opsX induced an approximately 7% decrease in xanthan production. The average amount of xanthan in the supernatant of the opsX mutant and wild-type cultures, measured at OD490, was 2.33 and 2.51, respectively (Fig. 4). In addition to their similar xanthan production, opsX mutant and wild-type strains showed similar growth patterns in YDC medium.
Because Kingsley et al. (1993) reported that mutation of opsX affects biosynthesis of LPS in X. campestris, we examined LPS in the wild-type and opsX mutant strains of Xoo. LPS extracts from the wild-type and mutant strains were prepared from whole-cell lysates and analyzed by SDS-PAGE. The silver-stained LPS gel is shown in Fig. 4. The ladder-like structure of E. coli LPS (Sigma-Aldrich) was used as a comparative sample. In these comparisons, the wild-type and mutant strains exhibited similar LPS banding patters (labeled I and II; Fig. 4).
Motility and growth rate
The motility of the wild-type and opsX mutant strains was investigated using semisolid swarm plates. In the swarm plate assay, we found that the opsX mutant strain was nonmotile compared with the wild-type strain. Following 11 days of incubation, the average diameter of opsX mutant colonies was zero cm, whereas that of wild-type colonies was 3.46 cm (Fig. 3F). These results supported the expression analysis showing downregulation of genes involved in Xoo chemotaxis due to mutation of opsX; consequently, the opsX mutant strain cannot maintain chemotaxis. We also compared the growth rates of the wild-type and opsX mutant strains (Fig. 3G). However, the growth rate of the opsX mutant was similar to that of the wild-type strain, indicating that mutation of opsX did not severely affect bacterial growth, although the growth rate of the opsX mutant was slightly increased (Fig. 3G).
Discussion
The opsX gene of Xoo showed a strong similarity (89.7% similarity at the amino acid level) to that of X. campestris pv. citrumelo strain 3048 and > 93% identity with genes from other xanthomonads (data not shown). In addition, the opsX gene showed strong conserved-domain homology with adenosine diphosphate (ADP)- heptose-LPS heptosyltransferase, which is involved in the biogenesis of the cell envelope of the bacterial outer membrane. Although Kingsley et al. (1993) reported that opsX is required for LPS assembly, virulence, and biosynthesis of EPS in X. campestris pv. citrumelo strain 3048, understanding the pleiotropic effects of this gene on the physiology of this bacterium remains a major challenge.
In the present study, the effects of mutation of opsX on xanthan, LPS production, and virulence, as well as on the expression of other genes, were evaluated in Xoo. Concordant with an earlier study by Kingsley et al. (1993), the opsX gene of X. campestris pv. citrumelo was found to affect biosynthesis of LPS and EPS. The M28 mutant (ΔopsX), derived from wild-type X. campestris pv. citrumelo strain 3048, lacked LPS and produced only 33% of the EPS produced by the wild type. However, the xanthan and LPS production changes observed in this study support the hypothesis that opsX is not required for xanthan and LPS production in Xoo (Fig. 4).
DNA microarray analysis revealed that several genes that play crucial roles in Xoo chemotaxis are downregulated by mutation of opsX (Fig. 3, Table S1). DNA microarray and enrichment analysis revealed that two other genes, flgE (encoding flagellar hook protein, XOO2572) and fliL (encoding flagellar protein, XOO2607), were also downregulated (Table 3). Consistent with the DNA microarray and quantitative RT-PCR analyses, the opsX mutant strain was nonmotile compared with the wild-type strain. No GO terms were found to be enriched among the upregulated genes, which indicates that genes that are typically downregulated played essential roles in virulence and bacterial chemotaxis.
LPS is a major component of the outer membrane of Gram-negative bacteria and is composed of three domains: 1) a hydrophobic anchor, known as lipid A; 2) a non-repeating oligosaccharide, known as the core region (an oligosaccharide consisting of an inner and outer region); and 3) a distinct repeating oligosaccharide, known as the O-antigen (Rick, 1987). In enteric bacteria, such as E. coli and Salmonella typhimurium, the inner core is composed of two ketodeoxyoctonate (KDO) units and two heptose units. The outer core region and the O-antigen are attached to one of the heptose units. In addition, several genes for synthesis of the inner core region have been identified from various enteric bacteria (Heinrichs et al., 1998; Schnaitman and Klena, 1993) and two genes including rfaC (waaC) and rfaF (waaF) were identified as heptosyl transferases for transfer of heptose from ADP-heptose to the inner core (Sirisena et al., 1992, 1994). Furthermore, Inoue et al. (2007) reported that all the genes that were involved in core oligosaccharide synthesis, but not those involved in O-antigen synthesis, were required for swarming and the inner core might be necessary for flagellar assembly and function of E. coli K-12 strain. As described above, BLASTP searches against the NCBI non-redundant database revealed that the opsX gene shared strong sequence similarity to ADP-heptose- LPS heptosyltransferase. This result indicates that the product of the opsX gene may also be responsible for the construction of the inner core of LPS as well as for flagellar assembly and function in Xoo.
Although further investigation is required to provide a better understanding of opsX and its role in bacterial chemotaxis, our findings are the first to suggest that mutation of opsX affects chemotaxis and bacterial motility, as well as virulence, in Xoo.
Supplementary Information
Acknowledgments
This work was supported by Konkuk University.
Notes
Articles can be freely viewed online at www.ppjonline.org.