Defense-Related Responses in Fruit of the Nonhost Chili Pepper against Xanthomonas axonopodis pv. glycines Infection
Article information
Abstract
Xanthomonas axonopodis pv. glycines (Xag ) is a necrotrophic bacterial pathogen of the soybean that causes bacterial pustules and is a nonhost pathogen of the chili pepper. In the current study, chili pepper fruit wound inoculated in planta with Xag 8ra formed necrotic lesions on the fruit surface and induced several structural and chemical barriers systemically in the fruit tissue. The initial defense response included programmed cell death of necrotizing and necrotized cells, which was characterized by nuclear DNA cleavage, as detected by TUNEL-confocal laser scanning microscopy (CLSM), and phosphatidylserine exposure on cell walls distal to the infection site, as detected by Annexin V FLUOS-CLSM. These two responses may facilitate cell killing and enhance transportation of cell wall materials used for cell wall thickening, respectively. The cells beneath the necrotic tissue were enlarged and divided to form periclinal cell walls, resulting in extensive formation of several parallel boundary layers at the later stages of infection, accompanying the deposition of wall fortification materials for strengthening structural defenses. These results suggest that nonhost resistance of chili pepper fruit against the nonhost necrotrophic pathogen Xag 8ra is activated systematically from the initial infection until termination of the infection cycle, resulting in complete inhibition of bacterial pathogenesis by utilizing organ-specific in situ physiological events governed by the expression of genes in the plant fruit organ.
Introduction
Plants have evolved a variety of barriers preformed or induced after pathogen infection to defend themselves. These defense structures and related materials inhibit pathogens from entering and spreading throughout the plant (Agrios, 2005; Yun and Loake, 2002). Host plants can be resistant to pathogens that are virulent to other host plants in the same species by R-gene-mediated resistance. This type of resistance is often governed genetically by one major gene that can be easily invalidated by pathogens newly evolved, with the corresponding avirulence (Avr) genes modified or removed (McDowell and Woffenden, 2003; Vera Cruz et al., 2000). Alternatively, non-host resistance is a broad-spectrum plant defense resulting in all members of a plant species being immune to all isolates of microorganisms pathogenic to other plant species, which is assumed to be controlled by multiple genes that are not easy to circumvent, thus providing durable disease resistance (Ham et al., 2007; Holub and Cooper, 2004; Senthil-Kumar and Mysore, 2013).
Programmed cell death (PCD) is a typical resistance response that results from host-parasite interactions of specific plant resistance and the pathogen’s avirulence genes, leading to a hypersensitive response (HR) (Greenberg et al., 1994; Heath, 2000; Jones and Dangl, 2006; Lord and Gunawardena, 2012). This is also a key feature of nonhost plant resistance at the initial inoculation site, especially against biotrophic pathogens whose survival and growth depend on utilizing living host plant tissue as a nutritional source (Hof et al., 2014). However, the resulting HR after PCD does little to affect necrotrophic pathogens that are capable of utilizing organic materials derived from decaying or dead cells. To combat these types of pathogens, a subsequent defense response is required to fully inhibit their infection process (Kim et al., 2004; Strange, 1998). This may also hold true for resistance of nonhost plants against nonhost necrotrophic pathogens.
Xanthomonas axonopodis pv. glycines (Xag) is a necrotrophic pathogen that causes a bacterial pustule in the soybean plant as well as large brown lesions forming pustules and ragging. In the nonhost chili pepper under natural conditions, Xag does not induce disease (Hwang and Lim, 1992; Park and Hwang, 1999). However, infiltration of Xag into nonhost plant leaf tissues leads to HR, which is related to the expression of HR and pathogenicity genes (hrp) (Park and Hwang, 1999). Chili pepper leaves infiltrated with Xag suspensions also form necrotic lesions, and electron microscopy of infected leaf tissues has been used to confirm subcellular changes related to HR. These are characterized by separation of the plasma membrane from the cell wall, formation of cell wall apposition, and cellular necrosis (Jeon et al., 2002). However, resistance responses other than HR have not been documented in the nonhost defense response of other organs of the chili pepper against necrotrophic pathogen infection. Therefore, the aim of this study was to investigate the initial and latestage histopathological responses of the chili pepper fruit to Xag infection to better understand the overall defense mechanisms of a nonhost plant in response to a necrotrophic bacterial pathogen.
Materials and Methods
Bacterial strain, host plant and inoculation
The bacterial strain used in this study was a wild type strain (8ra) of Xag (Xag 8ra) and had been used in a previous study (Jeon et al., 2002). Xag 8ra was grown on yeast extract calcium carbonate (YDC) agar at 28°C for 3 days prior to use. Chili pepper (Capsicum annuum cv. Bugang) plants were grown at 25°C in a greenhouse. Xag 8ra cells on YDC agar were suspended in sterilized distilled water (SDW) and adjusted to an optical density of 0.5 at 600 nm (approximately 2 to 5 × 108 cells/ml) using a Pharmacia Biotech Ultrospec 4000 spectrophotometer (Spectralab Scientific Inc., Markham, ON, Canada). Wound inoculation was performed in planta by rubbing the bacterial suspension-soaked sponge or the control SDW-soaked sponge onto pepper fruits attached to the plants immediately after pin-prick wounding (about 0.2 mm [depth] × 5.0 mm [length]). Inoculated pepper fruits were placed in a clean plastic bag in which SDW was sprayed to maintain 100% relative humidity inside. The inoculated and control plants were placed at 25 ± 2°C in a greenhouse. Symptom development was observed daily until 15 days after inoculation.
Confocal laser scanning microscopy (CLSM) for PCD in chili pepper fruit tissues infected with Xag 8ra
Two trademarks of PCD, nuclear DNA cleavage (damage) and phosphatidylserine (PS) exposure, at the early stage of PCD, were examined using the light microscopic TUNEL (terminal deoxynucleotidyl transferase-mediated dUTP nick end labeling) assay and the Annexin-V FLOUS (FITC-labeled Annexin V) staining assay, respectively (van Engeland et al., 1998; Wyllie, 1980). To detect nuclear DNA cleavage using the TUNEL assay, fruit sections were fixed in 4% paraformaldehyde in phosphate-buffered saline solution (PBS; 137 mM NaCl, 2.7 mM KCl, 10 mM Na2HPO4, 2 mM KH2PO4, pH 7.4) for 20 minutes, rinsed with PBS, then incubated in 0.1% Triton X-100 (in 0.1% aqueous sodium citrate) for 2 minutes at 2°C to 8°C on ice for permeabilization. Next, sections were rinsed twice with PBS and added along with 50 μl TUNEL reaction mixture to the sections and incubated at 37°C for 60 minutes in a humidified chamber. To detect PS exposure by Annexin-V FLOUS assay, fruit sections were fixed with a freshly prepared paraformaldehyde solution (4% in PBS) followed by 100 μl Annexin-V fluorescein solution (Roche Applied Science, Mannheim, Germany) in HEPES buffer and incubated at 15°C to 25°C for 10 to 15 minutes, followed by DNA staining of the sections for 10 to 15 minutes with propidium iodide, which is not permeable to live cells (Nicoletti et al., 1991). Following the TUNEL reaction and Annexin-V FLUOS staining, sections were rinsed three times with PBS and observed under a CLSM (Radiance-2000; Bio-Rad, Hertfordshire, UK) using an excitation wavelength in the range of 450 to 500 nm and an emission wavelength in the range of 515 to 565 nm.
Light microscopy (LM) and transmission electron microscopy (TEM) of chili pepper fruit tissues infected with Xag 8ra
Chili pepper fruit tissues infected with Xag 8ra were excised periodically and hand-sectioned with a razor blade. The sections were observed under a compound light microscope (Axiophot; Zeiss, Oberkochen, Germany) after staining with 0.1% toluidine blue O. Other infected tissues were excised and fixed in Karnovsky’s fixative in 0.05 M cacodylate buffer (pH 7.2) for 2 hours. The fixed fruit tissues were rinsed in the same buffer solution three times each for 20 minutes and post-fixed in 1% osmium tetroxide in the same buffer for 2 hours. The fixed samples were washed briefly in distilled water and stained en bloc with 0.5% uranyl acetate overnight. They were then dehydrated in an ethanol series (30%, 50%, 80%, 95%, and three times of 100%) each for 10 minutes and further dehydrated in propylene oxide for 15 minutes. The dehydrated tissues were embedded in Spurr’s epoxy resin (Spurr, 1969) and allowed to polymerize at 70°C for 8 hours in an oven. Semi-thin sections of about 1 to 2 μm thick were cut with a glass knife on an MT-X ultra-microtome (RMC Inc., Tucson, AZ, USA) and mounted onto glass slides. These semi-thin sections were stained with 0.1% toluidine blue O and observed under a compound light microscope (Axiophot). The embedded samples were sectioned at 80 to 90 nm thickness with a diamond knife on an MT-X ultra-microtome to make ultrathin sections, which were stained with 2% uranyl acetate and lead citrate for 7 minutes each and examined under a JEM-1010 electron microscope (JEOL Ltd., Tokyo, Japan) at an acceleration voltage of 80 kV. At least three samples were observed per treatment each time.
Deposition of defense-related materials in pepper fruit tissues infected with Xag 8ra
The deposition of three materials that are related to cell wall fortification was examined (Agrios, 2005). For this, chili pepper fruit tissues infected with Xag 8ra were excised and fixed in Karnovsky’s fixative for 2 hours. The fixed tissues were hand-sectioned and stained with phloroglucinol-HCl for lignin and sudan IV for cutin and suberin following procedures described previously (Jensen, 1962; Yeung, 1998). Lignin deposition was detected by its red under a compound light microscope (Axiophot), as described in the above reference (Jensen, 1962). For sudan IV staining, both cutinized and suberized cell walls were detected as red under a fluorescent microscope (Axiophot) using an excitation wavelength of 390 to 490 nm and a 515 nm barrier filter, thus differentiating the two materials from each other by their localization in the fruit tissues examined, and comprising the suberin deposition in lingo-suberized cell walls showing yellowish in color (Rittinger et al., 1987).
Results
Symptoms of Xag 8ra pepper fruit infection
Symptoms of bacterial infection by Xag 8ra on pepper fruit included the formation of blackish necrosis on the wounding lesions, which was initially confined to the lesion brims and later expanded inwardly with no or little outward lesion expansion until the end of the experiment (Fig. 1A). Conversely, the control wound-inoculated pepper fruits had only wound vestiges without necrotic lesions (Fig. 1B).
CLSM for PCD in fruit tissues infected with Xag 8ra
Two indicators of PCD, nuclear DNA cleavage and PS exposure, were detected using the TUNEL assay and Annexin-V FLUOS staining, respectively (Fig. 2). Pepper fruits without pathogen inoculation did not have TUNEL-positive signals in nuclei at the wound site or Annexin-V FLUOS-positive signals on plasma membranes (Fig. 2A, D). Likewise, these signals were absent immediately following pathogen inoculation (data not shown). TUNEL-positive nuclear signals were rarely observed in pepper fruit tissues by 12 to 18 hours after pathogen inoculation (data not shown), but were found extensively in epidermal and sub-epidermal cells in and around the inoculation sites by 24 hours after wound inoculation (Fig. 2B, C). By 16 hours after inoculation, there were numerous positive Annexin-V FLUOS staining signals along the cell walls, likely cell membranes, distal to the infection site (Fig. 2E), which was at an earlier time than TUNEL signals, were observed, and further increased by 24 hours after inoculation (Fig. 2F).
Confocal laser scanning microscopy of chili pepper fruit tissues with wounding alone (A, D) and inoculated with Xanthomonas axonopodis pv. glycines 8ra (B, C, E, F) followed by TUNEL (A–C) and Annexin-V FLUOUS staining (D–F). There were no positive signals on the fruit tissues underneath the wound site (WS) in the control inoculation (A, D), but TUNEL-positive signals are evident (arrows) and localized in the nuclei (N) mostly in epidermal and subepidermal cells 24 hours after inoculation (B, C), while Annexin-V FLUOUS-positive signals (arrows) localized on the cell walls distal to the inoculation site (IS) 16 hours (E) and 24 hours (F) after inoculation. (C) Enlarged image of the rectangular area in (B). Scale bars = 50 μm.
PCD-related structural changes of pepper fruit infected with Xag 8ra
LM and TEM of pepper fruit tissues inoculated with Xag 8ra identified structural features of PCD and HR, which were characterized by necrotic and necrotizing cells, accompanied by modified cells in the vicinity of the wound (Fig. 3). Using LM, we confirmed that pepper fruit tissues affected by infection contained necrotic or necrotizing cells and modified cells with vacuolated intact cytoplasm in their vicinity, often demarcated by necrotic cell walls (Fig. 3A). Using TEM, we observed that the necrotic and necrotizing cells were characterized by a shrunken degenerating cytoplasm separated from the degenerated necrotic cell walls (Fig. 3B). Nuclear features were homogeneously heterochromatic at 1 hours after inoculation (Fig. 3C), and by 24 to 60 hours after inoculation, had proliferated with aggregated heterochromatic materials distributed randomly in the nucleoplasm (Fig. 3D, E). Finally, 5 days after inoculation, the chromatin materials were dispersed in the shrunken necrotic cytoplasm that was separated from the degenerated cell walls (Fig. 3G).
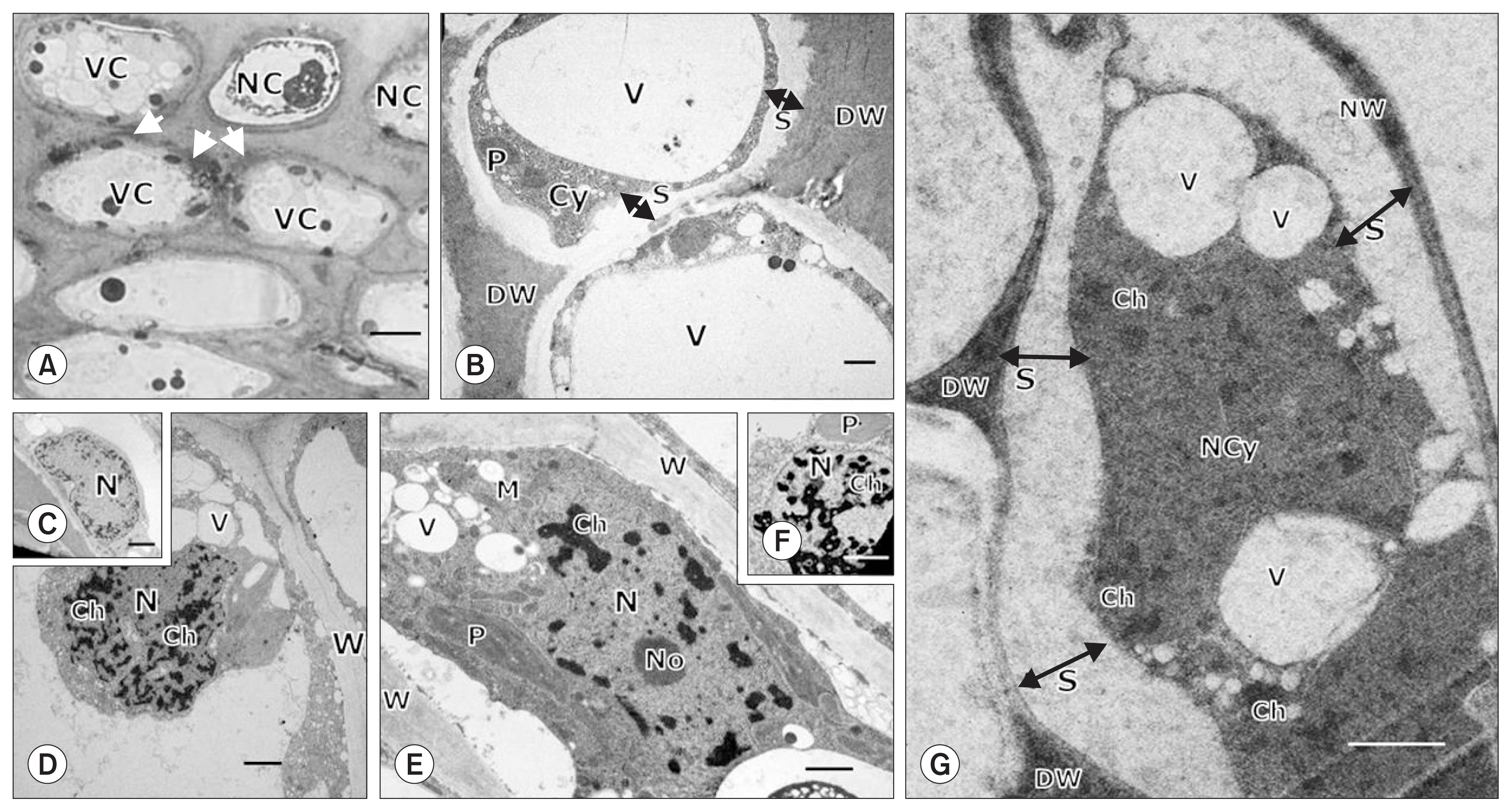
Light microscopy (A) and transmission electron microscopy (B–G) of chili pepper fruit tissues inoculated with Xanthomonas axonopodis pv. glycines 8ra showing structural changes related to hypersensitive and necrotic responses 3 days (A, B), 1 hour (C), 24 hours (D), 48 hours (E), 60 hours (F), and 5 days (G) after inoculation. The inoculated fruit tissue (A) contained necrotic or necrotizing cells (NC) along with highly vacuolated (VC) adjacent cells with necrotized cell walls (arrows). The necrotic or NC (B) were characterized by cytoplasmic shrinkage indicated by separation (S) of the cytoplasm (Cy) from the degenerated cell wall (DW). Initially (C), nuclear (N) features in the inoculated tissues were homogeneously heterochromatic. In later stages (D–G), these features continued to accumulate condensed heterochromatin materials (Ch) that were distributed in the nucleoplasm and finally dispersed in the necrotized cytoplasm (NCy) and in necrotizing (E, F) and necrotized cells (G). Cellular organelles such as plastids (P) and mitochondria (M) in (B), (E) and (F) appeared degenerated, indicating cytoplasmic degeneration. V, vacuole; W, cell wall; No, nucleolus; NW, normal intact cell wall; S, separation of cell wall from cytoplasm. Scale bars = 10.0 μm for (A) and 1.0 μm for (B) through (G).
Other initial defense-related structural changes at the inoculation site of pepper fruit
There were other structural changes that occurred at the inoculation site concurrent with PCD-like structural changes (Fig. 4). For instance, the cytoplasmic contents of cells adjacent to necrotic cells had increased numbers of vacuoles due to cytoplasmic infiltration, indicated by proliferous cytoplasmic fragmentations 3 days after inoculation (Fig. 4A). Around this time, some cells adjacent to the necrotic cells contained a dense cytoplasm with numerous small vacuoles, similar to meristematic cells, likely derived from the large vacuoles breaking down due to the infiltration of increased cytoplasmic contents (Fig. 4B). By LM, we observed that the affected cells bounding with and separated by the thickened necrotic cell wall and middle lamella from the necrotic cytoplasm contained an intact cytoplasm with the parallel cell walls, which were often significantly thickened (Fig. 4C). Likewise, by TEM, we found that numerous small vacuoles were produced in the intact dense cytoplasm, which appeared to be incorporated into the thickened cell wall, probably depositing cell wall materials onto the cell wall to increase cell wall thickness (Fig. 4D). At later stages of infection, these structural changes resulted in confinement of the bacterial cells in lysigenous intercellular spaces surrounded by thickened cell walls that were separated from the necrotic cytoplasm, consequently protecting the adjacent and bounding cells from degeneration (Fig. 4E).
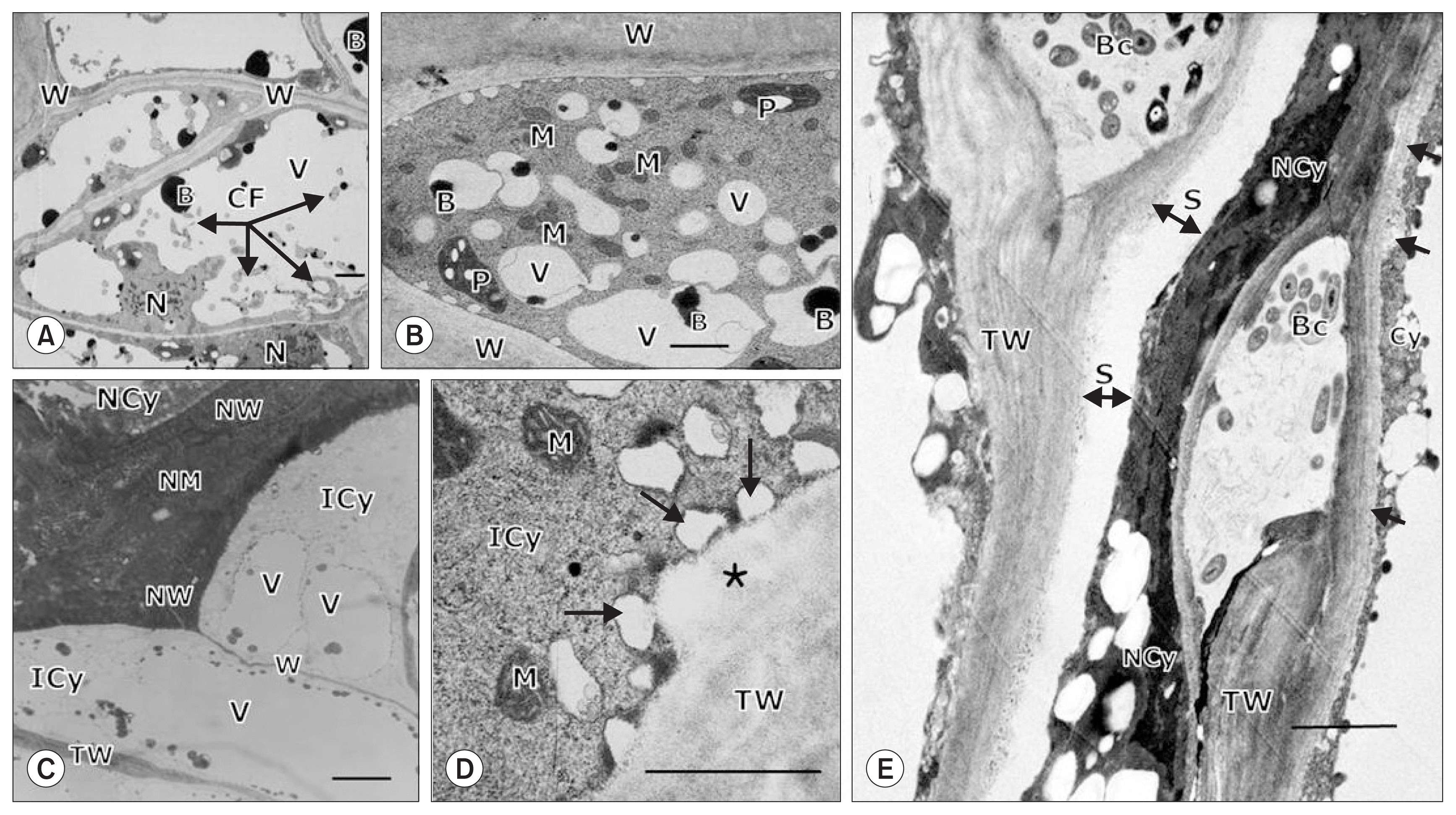
Light microscopy (A) and transmission electron microscopy (B–E) of chili pepper fruit tissues inoculated with Xanthomonas axonopodis pv. glycines 8ra showing structural features of cells bounding with necrotic cells with a necrotized cytoplasm (NCy) and walls (NW) and middle lamellae (NM) 3 days (A–D) and 15 days (E) after inoculation. The cells (A) bounding with the necrotic cell contained an intact cytoplasm (ICy) with parallel thin (W) and thick cell walls (TW), around which the incorporation of vesicles (arrows) likely containing cell wall materials into the cell wall to increase cell wall thickening (asterisk) in (B) was observed. The cytoplasms of the cells adjacent to the necrotic cells became dense with cytoplasmic infiltration into the vacuole (V), as indicated by cytoplasmic fragmentation (CF) (C), resulting in a dense cytoplasm with numerous small vacuoles like meristematic cells (D). In (E), structural features at a later stage of infection are evident, showing the freely gathered bacterial cells (Bc) confined in intercellular spaces surrounded by thickened cell walls (TW) that were separated (S) from the NCy, thus protecting the cytoplasm (Cy) from degeneration. In (E), additionally thickened cell walls are noted by arrows. B, inclusion body; M, mitochondria; P, plastids. Scale bars = 5.0 μm for (A) and 2.0 μm for (B) through (E).
Later defense-related structural changes and deposition of cell wall fortification materials in pepper fruit tissues infected with Xag 8ra
Following control wound inoculation, there were no subsequent defense-related structural changes observed in fruit tissues (data not shown). Similarly, one day after wound inoculation, there were no defense-related structural changes around the tissue necrotized by the pathogen infection (Fig. 5A). However, 4 days after inoculation, the cells beneath the necrotic tissue were enlarged with few cell divisions (Fig. 5B), and the cell divisions occurred in a series of cells, forming paralleled periclinal walls. By 8 days after inoculation, a layer had formed (Fig. 5C). Later, by 15 days after inoculation, several paralleled boundary layers were extensively formed beneath the necrotic tissue (Fig. 5D), which were found to auto-fluoresce using a fluorescent microscope (data not shown). The cell walls at the bottom of the inoculated wound had accumulated cell wall fortification materials 4 days after inoculation (Fig. 5E, G), and by 15 days following inoculation, this accumulation had become extensive. We also observed that the tissue above the necrotic tissue became lignosuberized (i.e., lignin and suberin containing), and the lower cutinized tissue was bounded underneath with intact tissue (Fig. 5F, H).

Light microscopy (A–F) and fluorescent microscopy (G, H) of chili pepper fruit tissues one day (A), 4 days (B, E, G), 8 days (C), and 15 days (D, F, H) after inoculation with Xanthomonas axonopodis pv. glycines 8ra. In (A) to (D), the formation of defense structures (boundary layers, BL) is evident, and in (E) and (F), deposition of cell wall fortification materials such as lignin (Lig), and in (G) and (H), suberin (Sub) and cutin (Cut) are present in or under the necrotic tissue (NT) of the inoculation site (IS). The circled area in (B) is to highlight longitudinally enlarged cells in the process of cell division to form boundary layers. Scale bars = 50 μm.
Discussion
The phenomenon that most plants typically show resistance to the majority of pathogens is referred to as nonhost resistance, which is governed by a broad range of mechanisms regulated by multiple genes (Ham et al., 2007; Heath, 2000; Mysore and Ryu, 2004; Senthil-Kumar and Mysore, 2013). In the current study, chili pepper fruit (nonhost of Xag) wound-inoculated with Xag 8ra, a necrotrophic bacterial pathogen, formed necrotic lesions and developed several structural and chemical barriers to Xag 8ra infection. These defenses initially included PCD and HR, and later, boundary layer formation accompanied by cell wall thickening and deposition of cell wall fortification materials. These defense mechanisms are contrary to those employment against biotrophic pathogens, which are largely due to PCD in the infected tissue of the host plant (Glazebrook, 2005).
A key feature of nonhost plant resistance is PCD at the initial inoculation site, and is characterized by nuclear DNA cleavage and PS exposure (Hof et al., 2014; van Engeland et al., 1998; Wyllie, 1980). The nuclear degeneration caused by chromosomal DNA cleavage is one way to facilitate cell killing in PCD, accompanied by chromatin condensation, nuclear pycnosis and DNA fragmentation, consequently resulting in apoptotic body formation (Granville et al., 1998; Parrish and Xue, 2006; Wyllie et al., 1980). In the current study, TEM (Fig. 3D–G) was used to identify nuclear features characteristic of PCD such as chromatin condensation and nuclear pycnosis in dying cells (Elmore, 2007), as well as dispersion of chromatin materials in the dead cells of the chili pepper fruit, suggesting nuclear degeneration leading to cell death. It has been suggested that nuclear degeneration indicated initially by homogeneously heterochromatic nuclei and later by the dispersion of chromatin materials in the cytoplasm is indicative of syncytial cell degeneration in soybean roots infected with the soybean cyst nematode (Kim et al., 1987, 2010). Nuclear modifications and structural changes related to HR are also observed in resistant chili pepper fruit infected with the anthracnose pathogen (Kim et al., 2004). Together, these observations support the premise that nuclear DNA cleavage in cells with PCD is an efficient way to kill cells.
Contrary to the function of nuclear DNA cleavage in PCD, PS exposure on the outer plasma membrane leaflet (via loss of phospholipid asymmetry) in mammalian cells is responsible for signaling the PCD process to the environment, resulting in the removal of dead cell materials often by phagocytic uptake of macrophages (Fadeel and Xue, 2009; van Engeland et al., 1997, 1998). However, the role of PS exposure is not well-documented in PCD of plant cells, although the regulation of transmembrane bilayer lipid asymmetry is important in plant cell physiology, such as adaptation of plants to cold (Gomès et al., 2000).
In the current study, we used LM with Annexin-V FLUOS or toluidine blue O staining and TEM to evaluate Xag 8ra-infected pepper tissues. Our results demonstrate that PS exposure occurred mainly at the cell walls distal to the infection site, bounding occurred with thickened necrotized cell walls, and the middle lamella adjacent to modified cells with an intact cytoplasm contained numerous small vacuoles and thickened cell walls to which vesicles were incorporated at subsequent infection stages (Fig. 2E, F, 4A–D). These thickened cell walls may play a key role in the structural defense against pathogen invasion into the inner pepper fruit tissues as well as inhibit virulence factors transferred into adjacent healthy cells. These observations also suggest that the contents of apoptotic fruit cells may not be eliminated totally as in mammalian cells, but instead may be partially used for secondary cell wall thickening being fortified by the deposition of defense-related materials such as lignin, suberin and cutin, resulting in shrunken cellular remains that are condensed in lysigenous intercellular spaces (as shown in Fig. 4). Furthermore, the loss of membrane asymmetry caused by PS exposure may enhance the transportation of primers for cell wall materials through the plasma membrane to the cell wall where polymerization with their primers occurs (Kolattukudy, 1996). Therefore, the membrane alterations in pepper fruit infected with Xag 8ra may not only be related to PCD processes but also to preliminary factors for the formation of structural and chemical barriers to inhibit the spread of disease.
In host plants, wounding stimulates the formation of boundary layers such as the wound periderm and abscission layer as well as the deposition of lignin and suberin into boundary layers; however, these formations are enhanced significantly following inoculation of incompatible hemibiotrophic fungal pathogens (Kim and Kim, 2002; Kim et al., 2004, 2008, 2009; Rittinger et al., 1987). In the current study, the nonhost chili pepper plant had boundary layer formations that were remarkably enhanced following inoculation of the nonhost necrotrophic pathogen Xag 8ra. Enhancement of these structural defenses in the chili pepper fruit may be due to a residual effect of prior PCD responses, especially the PS exposure that leads to enhanced cell wall formation and fortification, as discussed above. This may also be true for enhanced structural changes observed at the later stages of the incompatible host-pathogen interactions mentioned above, suggesting there may be no fundamental phenotypic differences in inhibiting the infection processes of necrotrophic (or hemibiotrophic) pathogens in nonhost plants and incompatible host plants during the later stages of infection.
Nonhost plants may defend themselves against bacterial foliar pathogens by restricting the initial three-step infection process. This process begins with the initial pathogen entry, then multiplication in the apoplast and finally direct interaction with plant cells in which HR and HR-related defenses are induced in the cytosol, nucleus, plasma membrane, and cellular organelles (Senthil-Kumar and Mysore, 2013). Ultrastructural changes related to HR have been observed in pepper leaf tissues infected with Xag, but no boundary layer was formed to inhibit further pathogen invasion; however, bacterial cells were embedded in wall materials and cemented to plant cell walls at early infection stages (Jeon et al., 2002), which implies that inhibition for further bacterial invasion is not required. In contrast to these observations, in the current study, Xag cells gathered together within the intercellular spaces derived from cellular breakdown, but no bacterial cells were embedded in wall materials that were cemented to the cell wall to immobilize (Fig. 4E). This indicates that bacterial cells can activate the infection process, including pathogen invasion, for which additional defenses must be required to inhibit. These different defense responses in pepper fruit and pepper leaves may be related to differences in physiological incompatibility governed mostly by preformed defense structures as well as antimicrobial substances and/or nutrients secreted onto plant organ surfaces and in apoplastic regions (Chen et al., 2010; Cowan, 1999; Fan et al., 2011; Melotto et al., 2008; Rico and Preston, 2008; Senthil-Kumar and Mysore, 2013; Wang et al., 2012). The development of different organs and tissue types is governed by the selective expression of distinct fractions of the genome, resulting in the physiologically incompatible differences between the pepper fruit and pepper leaf (Ma et al., 2005). Plant organs that are known to form boundary layers in response to necrotrophic pathogen infections are chili pepper fruit (Kim et al., 2004, 2008), ginseng root (Jeon and Kim, 2008; Kim et al., 2009), cactus stem (Kim and Kim, 2002), and peach bark (Biggs, 1989), all of which are secondarily grown organs in which meristematic cells are regenerated to produce the secondary state of tissues (e.g., periderm) related to the boundary layers (e.g., wound periderm) (Biggs, 1986; Esau, 1977). All of these observations suggest that the nonhost defense response of the chili pepper fruit is dynamic and utilizes physiological events in situ that are governed during organ development by the enhanced expression of genes, probably for pathogenesis-related proteins that are required for PCD (Kim and Hwang, 2015) and key enzymes in cell wall-building processes, including lignin formation (Taheri and Tarighi, 2011, 2012).
In conclusion, nonhost resistance of the chili pepper fruit against Xag 8ra infection is activated systematically from the initial infection until termination of the infection cycle, initially inducing PCD and PCD-related defense structures leading to HR and cell wall thickening due to nuclear aberration and PS exposure, respectively, and later in the infection process inducing boundary layer formation beneath the infection site as a structural barrier against further pathogen invasion. Defense-related materials such as lignin, suberin and cutin were deposited onto the thickened and necrotized cell walls, thereby strengthening the cell wall structures. It is probable that this systematic resistant response may be required for the chili pepper fruit, a complicated plant storage organ with comparably lower physiological activity, to resist infections by necrotrophic bacterial pathogens that benefit from cell death.
Notes
Articles can be freely viewed online at www.ppjonline.org.
